









| Still deciding? Get samples of $ ! US$ 0.13/Piece Request Sample |
| Model NO. | USP2-USP5/0 | Sterilization | Ethylene Oxide (EO) |
| Guarantee Period | 3 Years | Product Color | Blue / Black |
| Product Structure | Non-Absorbent Monofilament | Material | Polyamide 6 - 6.6 |
| Packaging | Single Aluminum Foil Bag | Transport Package | 12 PCS/Box, 600 PCS/Carton |
| Production Capacity | 50,000 PCS/Day | Certificates | CE, ISO 13485, FSC |
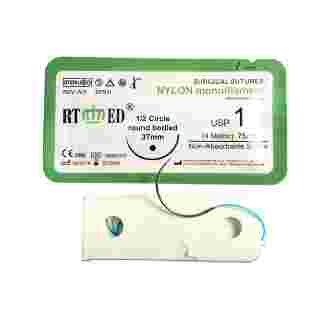
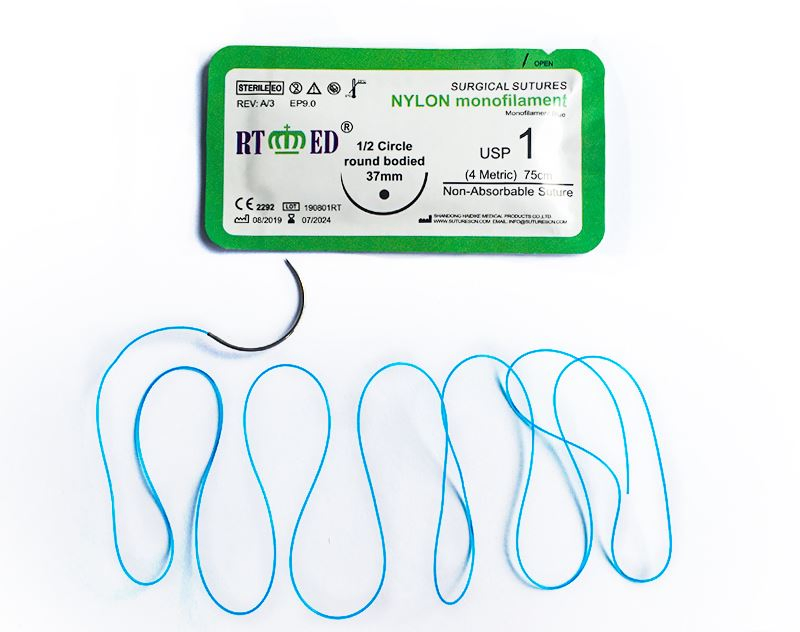
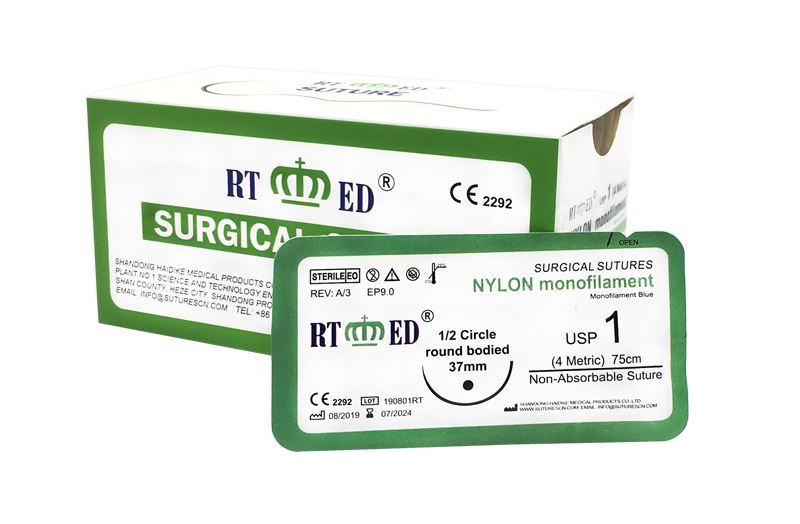
Nylon suture is a monofilament surgical suture synthesized by polyamide 6 and/or polyamide 6.6. As a non-absorbable suture, it is available in blue or black colors. Sterilized by ethylene oxide gas, the sizes range from USP 12/0 to USP 4. Nylon suture meets all requirements of the USP for non-absorbable surgical sutures.
This suture features high tensile strength, excellent compatibility, and low tissue reaction. Being a monofilament, it significantly reduces the risk of bacterial harboring and infection, ensuring no capillarity occurs during use.
The non-destructive suture needle produced by advanced drilling technology realizes the integration of needle and thread. This ensures the suture penetrates smoothly with minimal tissue damage. It is highly suitable for eye surgery, orthopedic surgery, and skin closure.


Nylon Sutures are indicated for use in general surgery. They are suitable for soft tissue ligation, including:
Note: Nylon Sutures should not be used where permanent retention of tensile strength is required for central neural or cardiac systems (e.g., artificial heart valves).
Our production facility specializes in medical consumables, utilizing high-quality stainless steel (300SS and 400SS Series) for surgical needles. We maintain rigorous quality control systems including ISO13485, CE, and FDA certifications. Our products are exported to more than 70 countries and regions worldwide, including Asia, Europe, Latin America, and the Middle East.





 Wigivida Medical
Wigivida Medical