
1 / 5










| Still deciding? Get samples of $ ! US$ 0.66/Piece Request Sample |

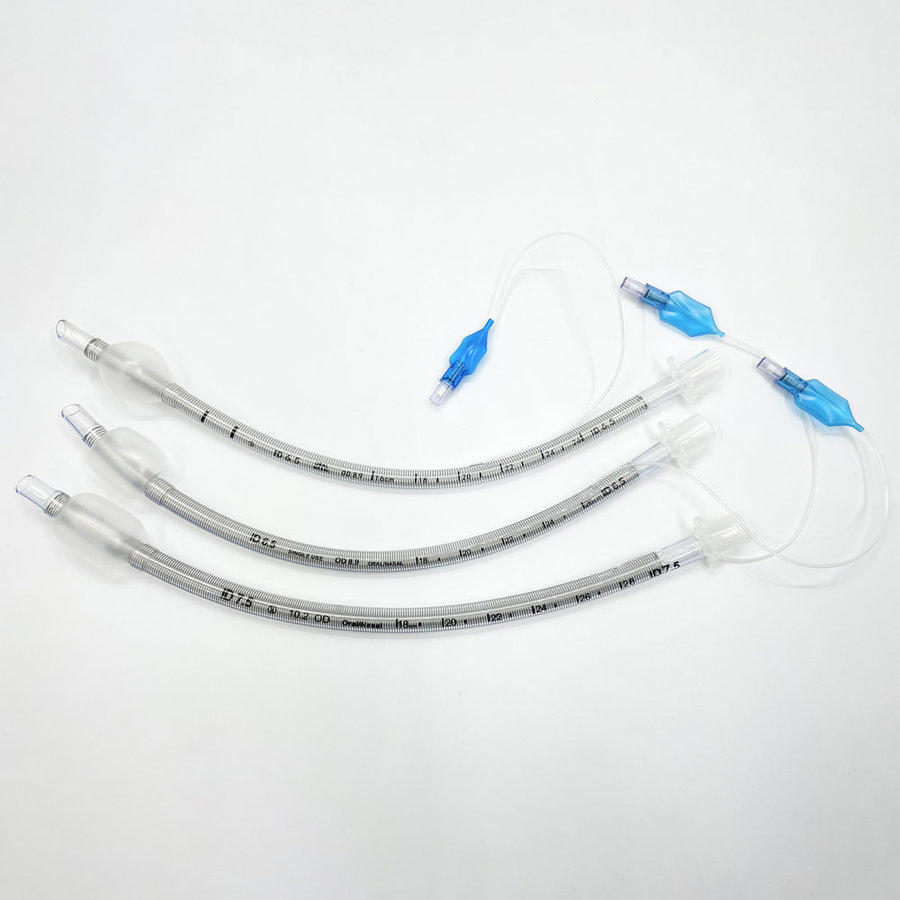
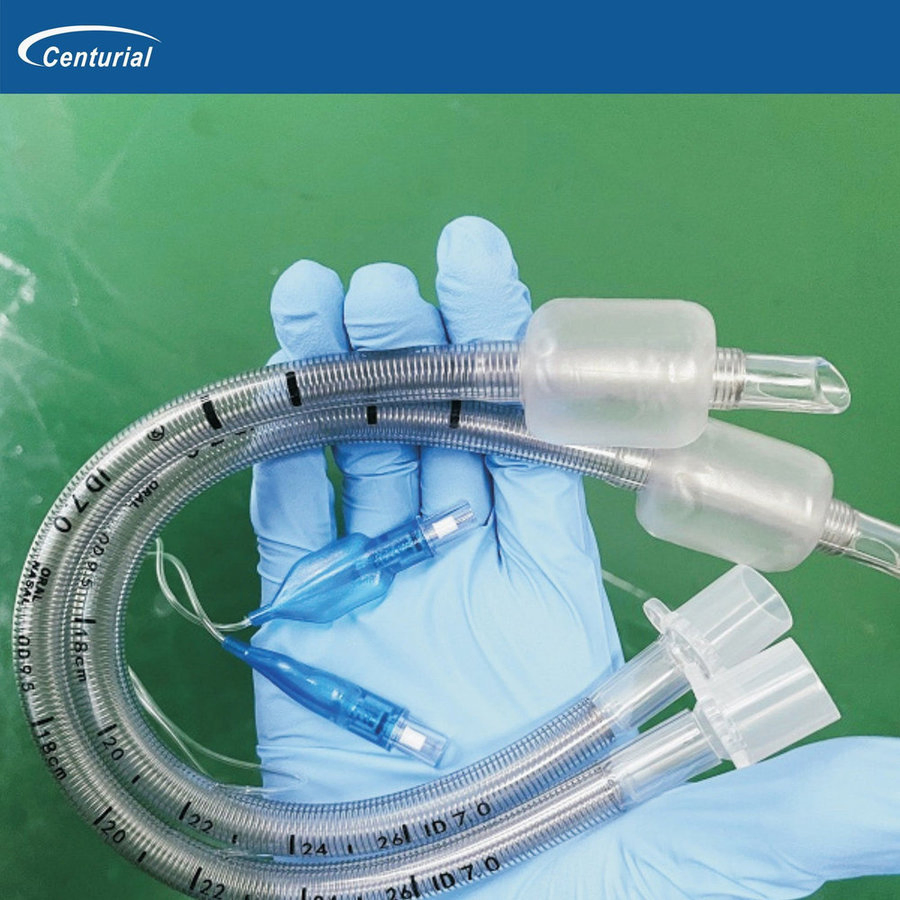
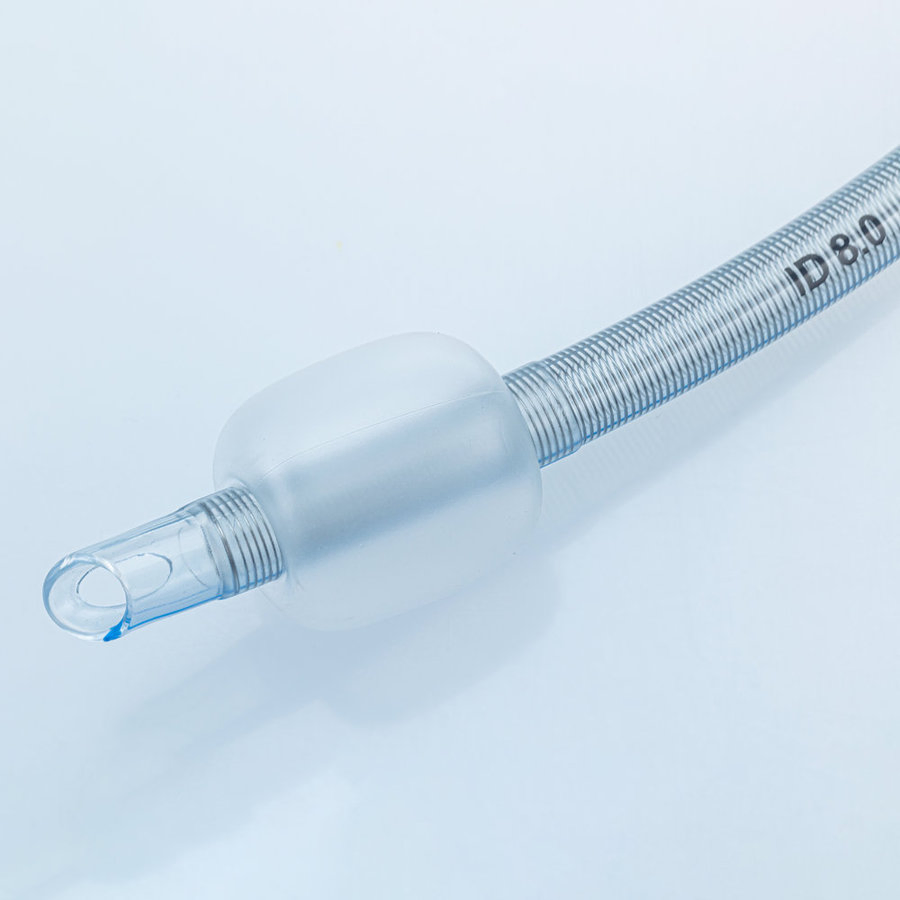
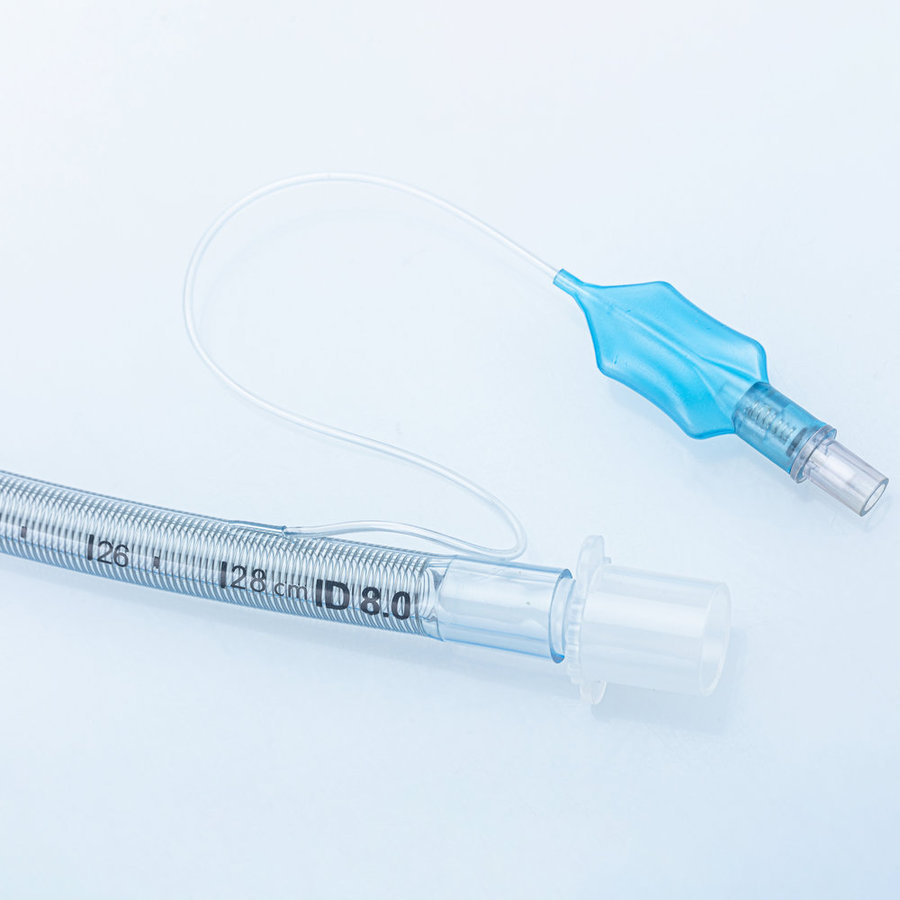
ENDOTRACHEAL TUBE (ET Tube / Tracheal Tube / ETT)
The Endotracheal Tube is a disposable medical device designed to establish and maintain a safe artificial airway during anesthesia and emergency resuscitation. Each tube is individually ETO-sterilized and packed, meeting international quality requirements for hospitals and clinics worldwide.



| Item No. | Size (I.D.) | Length (mm) | Item No. (Reinforced) | Size (I.D.) | Length (mm) |
|---|---|---|---|---|---|
| CT-ET01 | 2.0mm | 140±3.0 | CT-RE01 | 3.0mm | 174±5.0 |
| CT-ET02 | 2.5mm | 145±3.0 | CT-RE02 | 3.5mm | 191±5.0 |
| CT-ET03 | 3.0mm | 165±3.0 | CT-RE03 | 4.0mm | 211±5.0 |
| CT-ET04 | 3.5mm | 185±3.0 | CT-RE04 | 4.5mm | 240±5.0 |
| CT-ET05 | 4.0mm | 210±3.0 | CT-RE05 | 5.0mm | 256±5.0 |
| CT-ET06 | 4.5mm | 225±3.0 | CT-RE06 | 5.5mm | 286±5.0 |
| CT-ET10 | 6.5mm | 295±3.0 | CT-RE10 | 7.5mm | 211±5.0 |
| CT-ET14 | 8.5mm | 330±3.0 | CT-RE14 | 9.5mm | 211±5.0 |









 Wigivida Medical
Wigivida Medical