











| Still deciding? Get samples of $ ! US$ 0.05/Piece Request Sample |

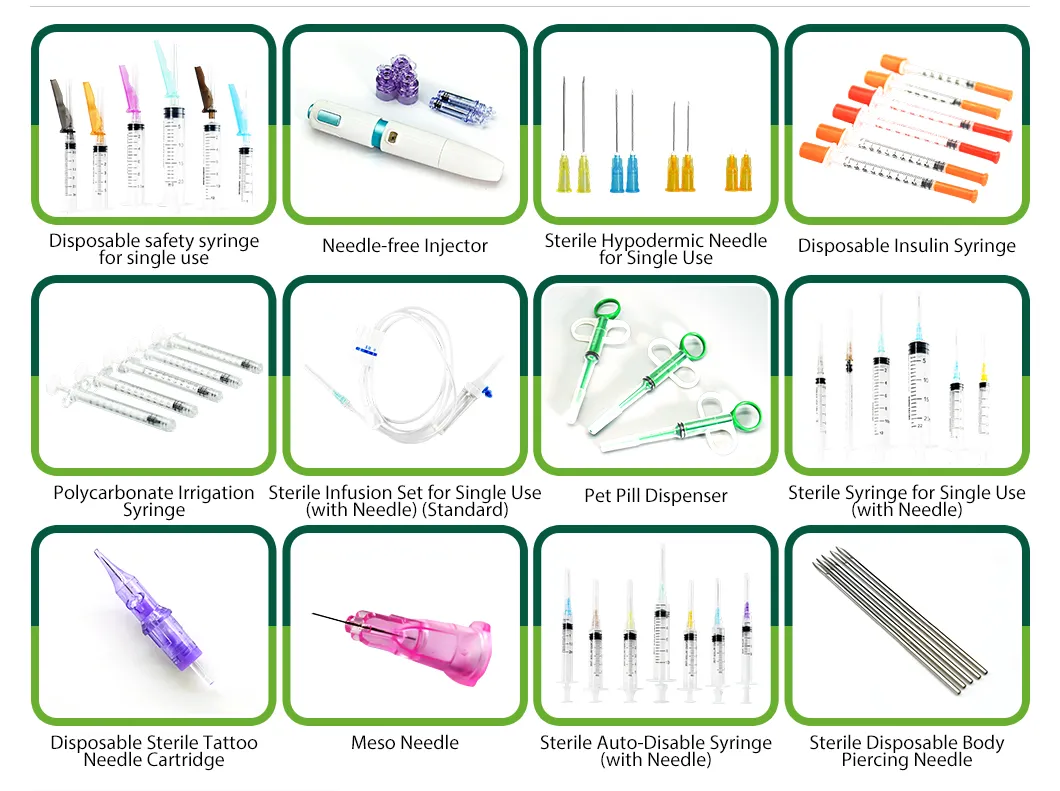

Established in 2005, we are a manufacturer specialized in disposable sterile medical consumables and related single-use devices. We focus on consistent quality, scalable production, and reliable delivery for global OEM/ODM customers. Our site covers approximately 32,000 m2 with a built-up area of over 60,000 m2. We operate more than 12,000 m2 of ISO Class 8 (Class 100,000 equivalent) cleanroom facilities.
Sterilization is supported by 8 EO sterilization cabinets. For quality assurance, we run an in-house testing center capable of biological, chemical, and physical testing, continuously investing in automation with 160+ sets of production equipment and 70+ sets of testing/QC equipment.


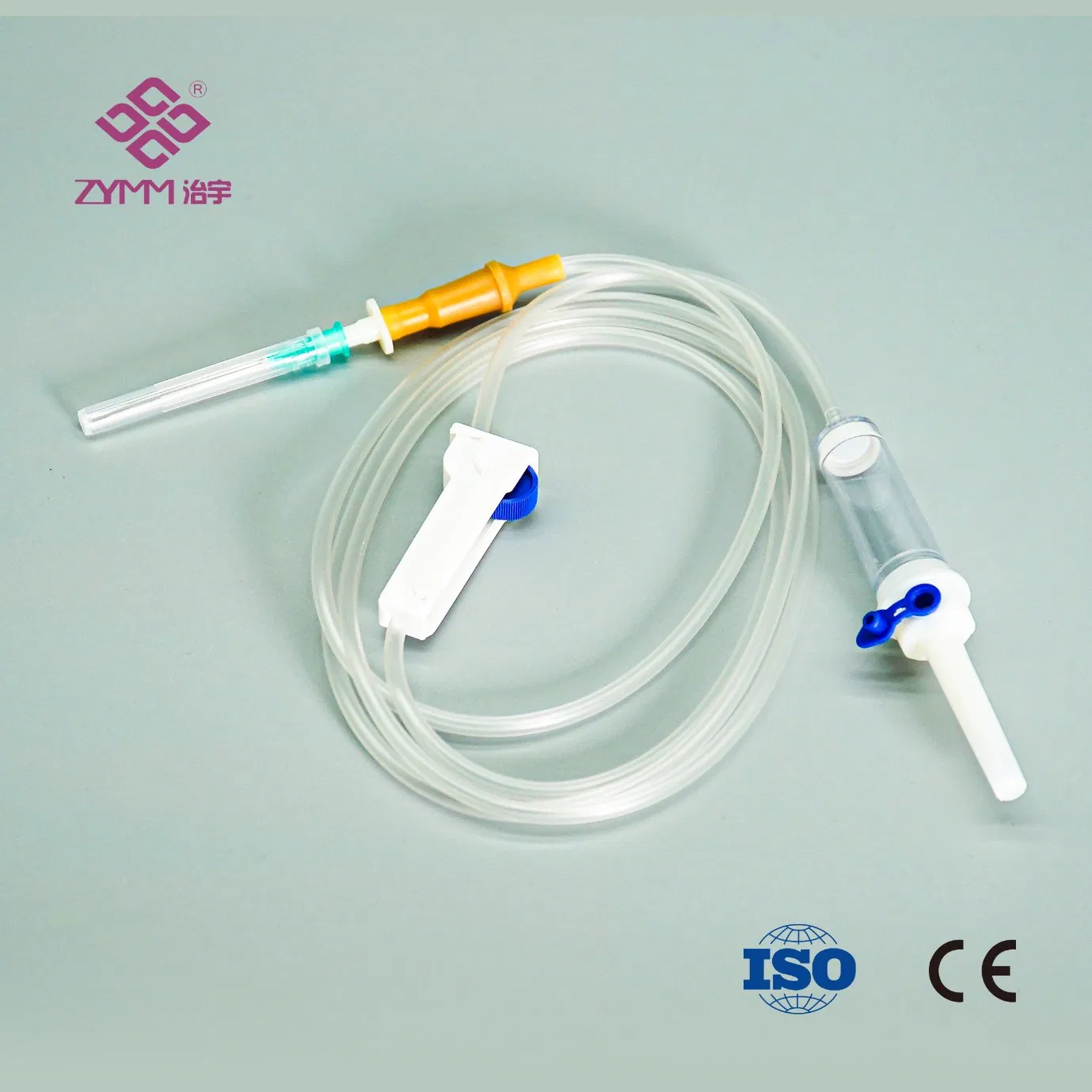
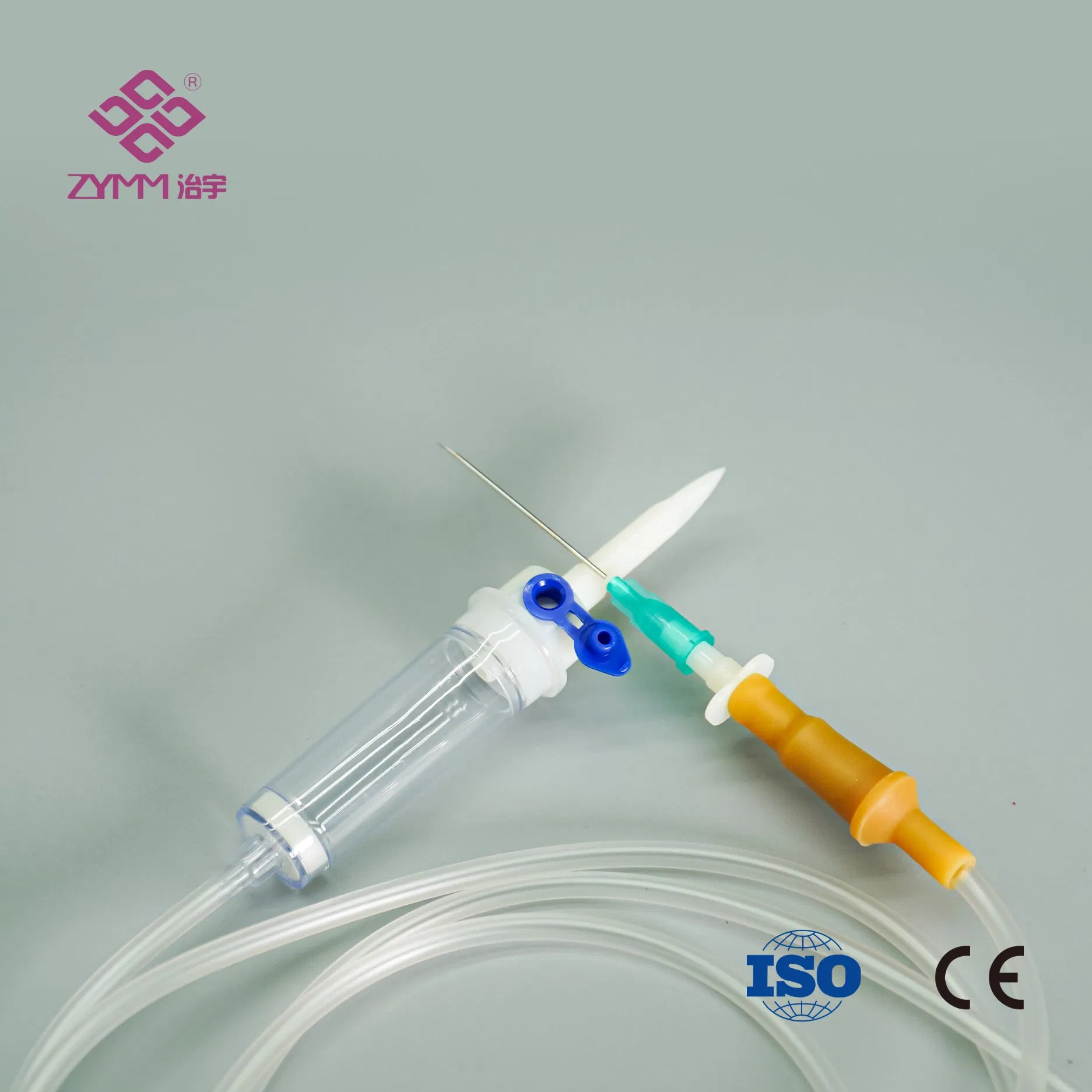
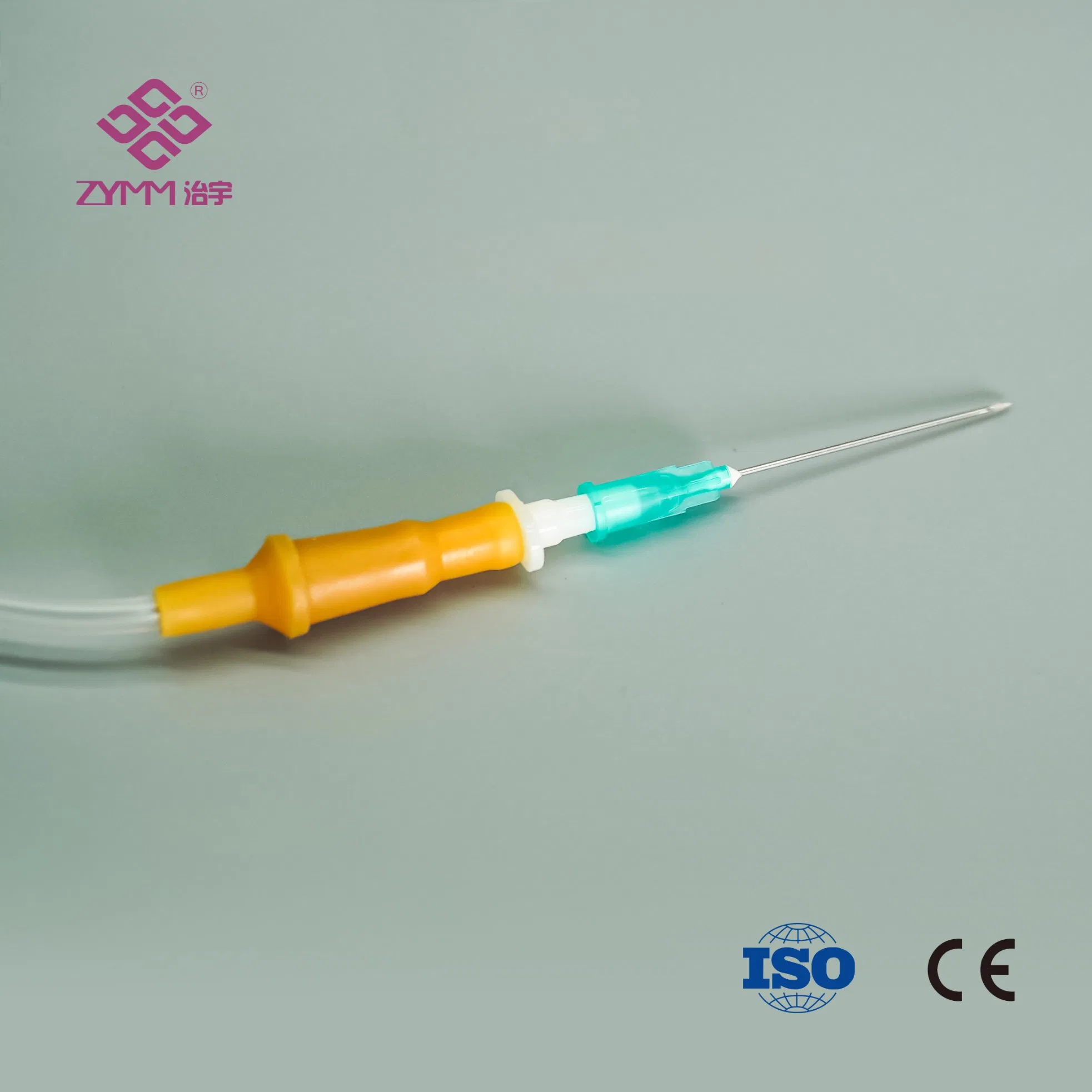
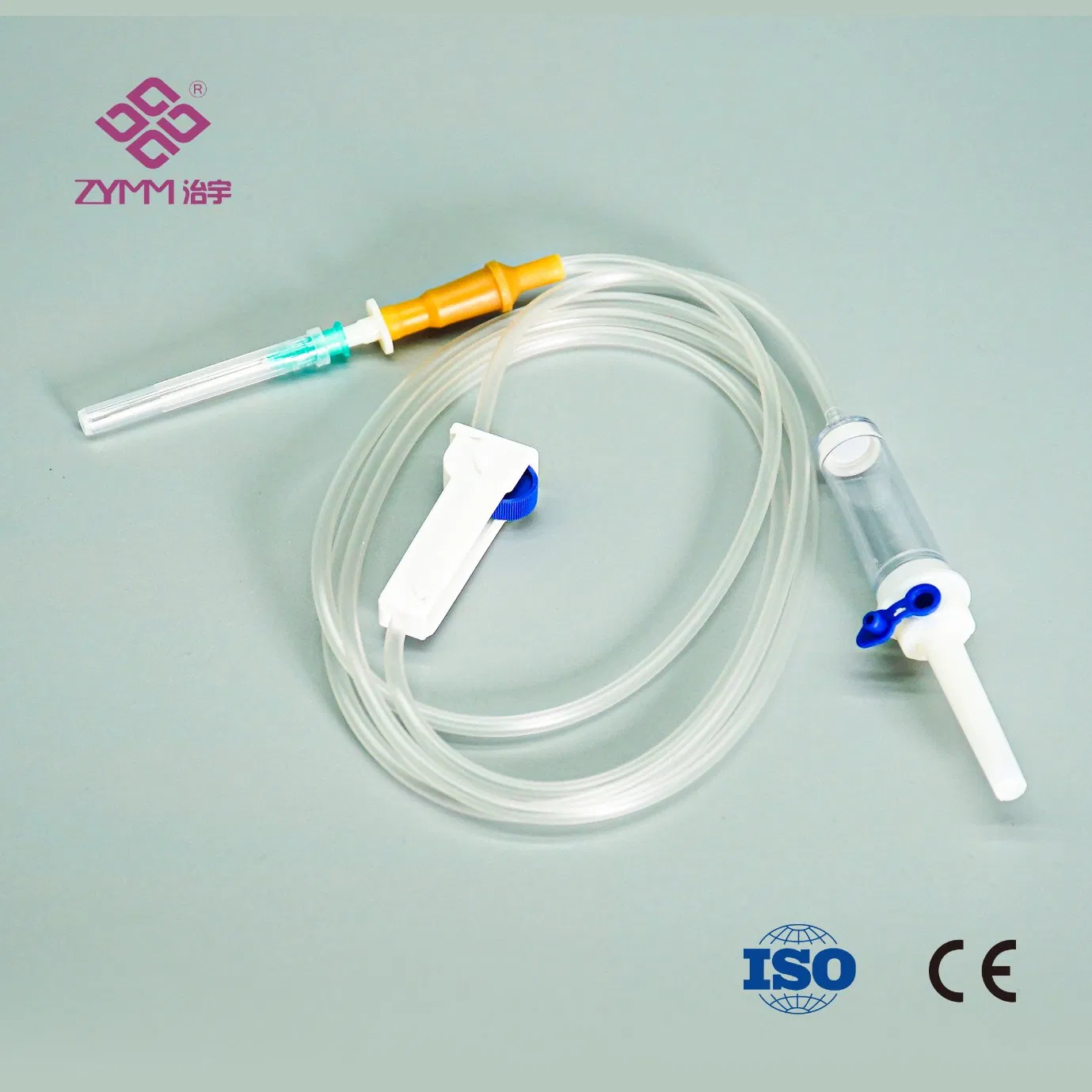
Our annual output of syringes exceeds 1 billion pieces, and infusion sets reach around 100 million pieces. Quality and compliance are core to our operations. We operate an ISO 13485 certified quality management system and hold EU MDR (EU) 2017/745 QMS certificates. We support customers with market-specific documentation, private labeling, and customized configurations.



We provide export-ready packing options including PE pouches or blister packaging. We work closely with customers on shipping marks, palletization, and mass production scheduling to ensure reliable lead times.
 Wigivida Medical
Wigivida Medical