
1 / 5










| Still deciding? Get samples of $ ! US$ 1/Piece Request Sample |
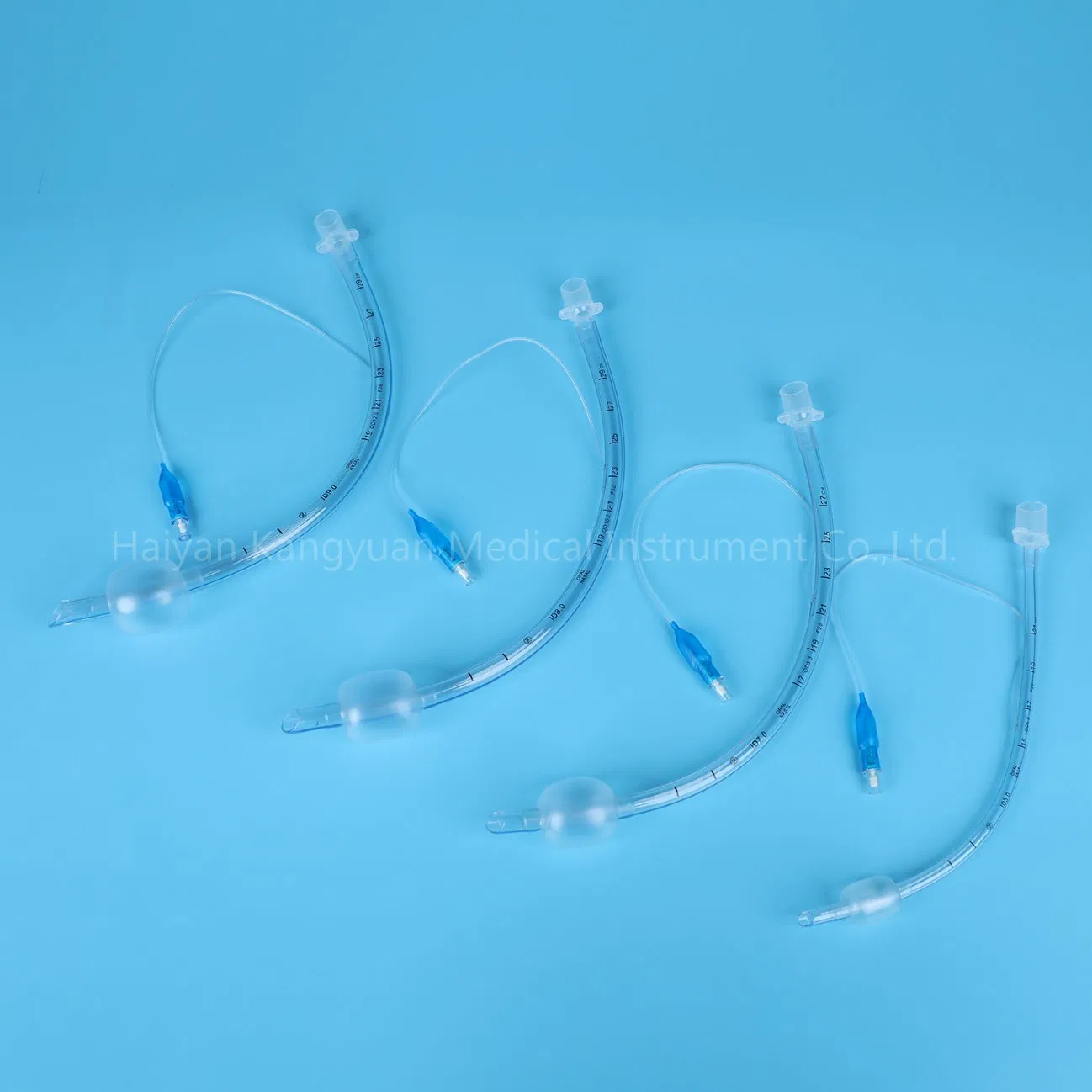
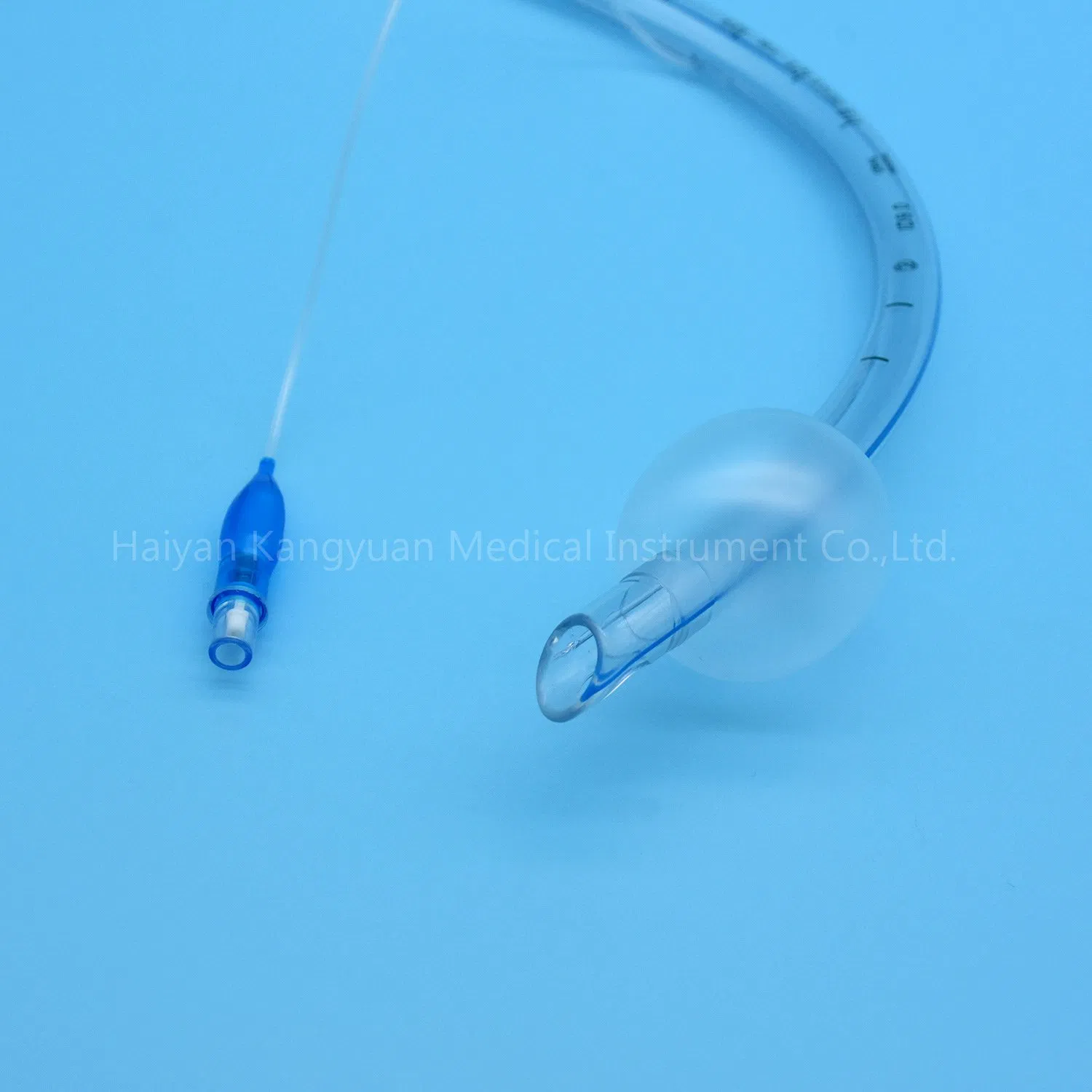
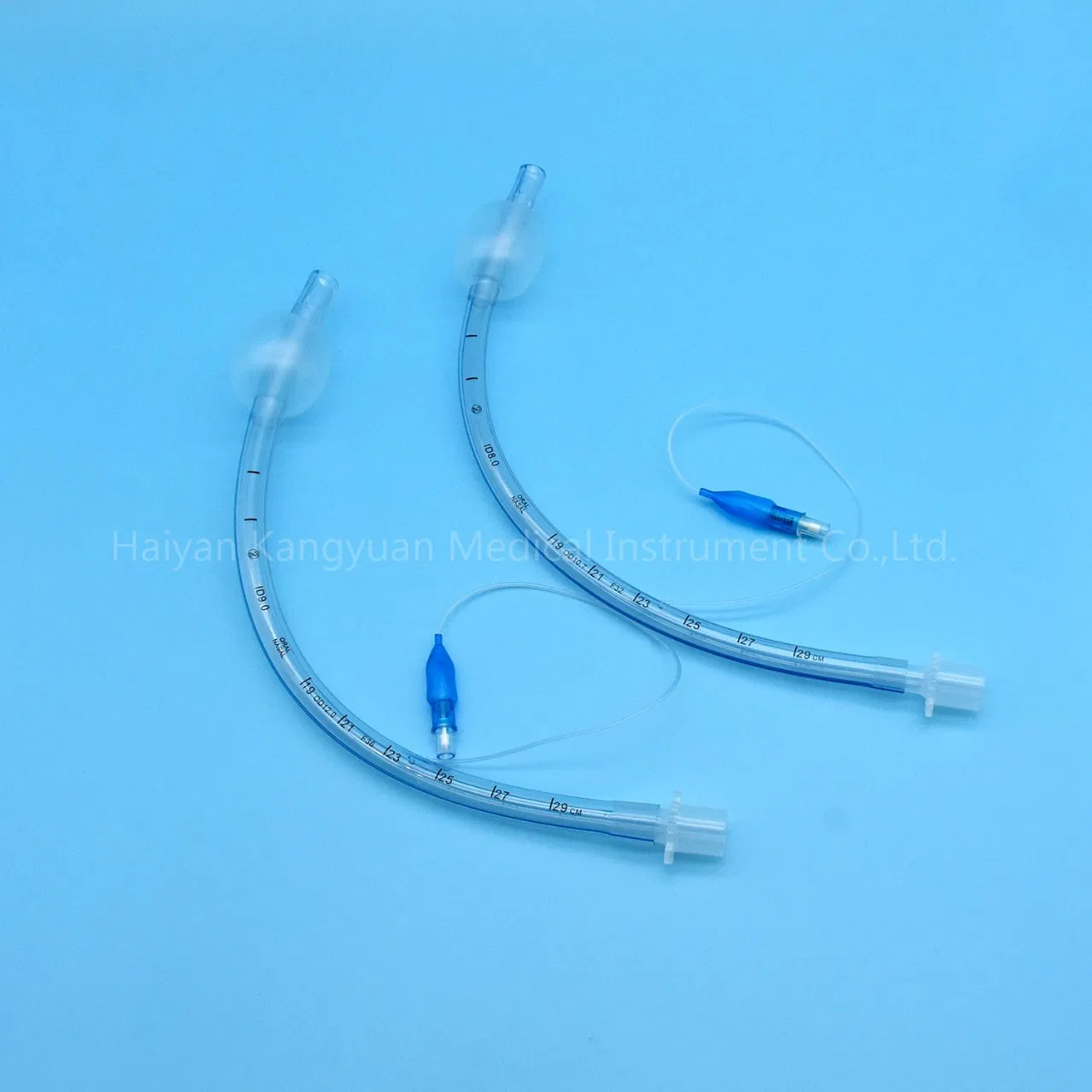
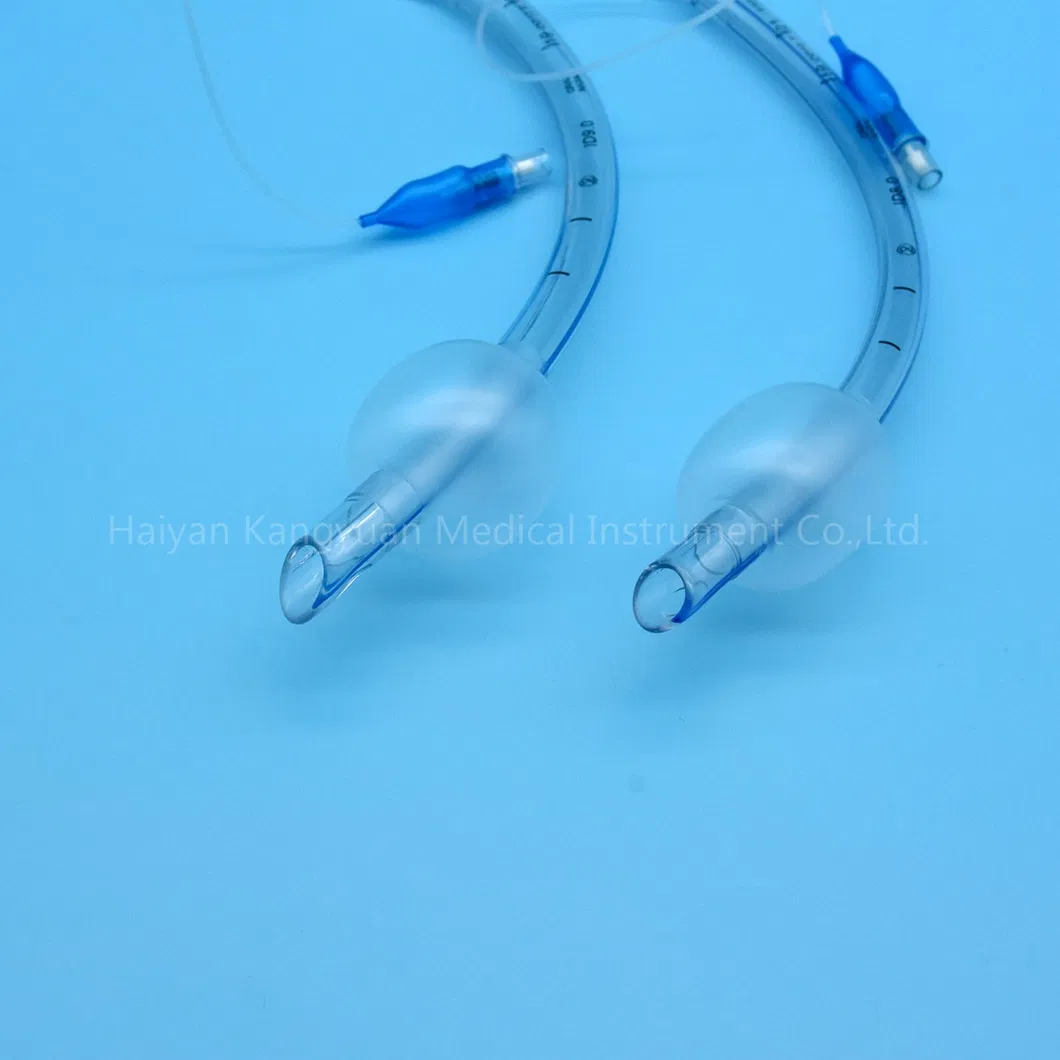
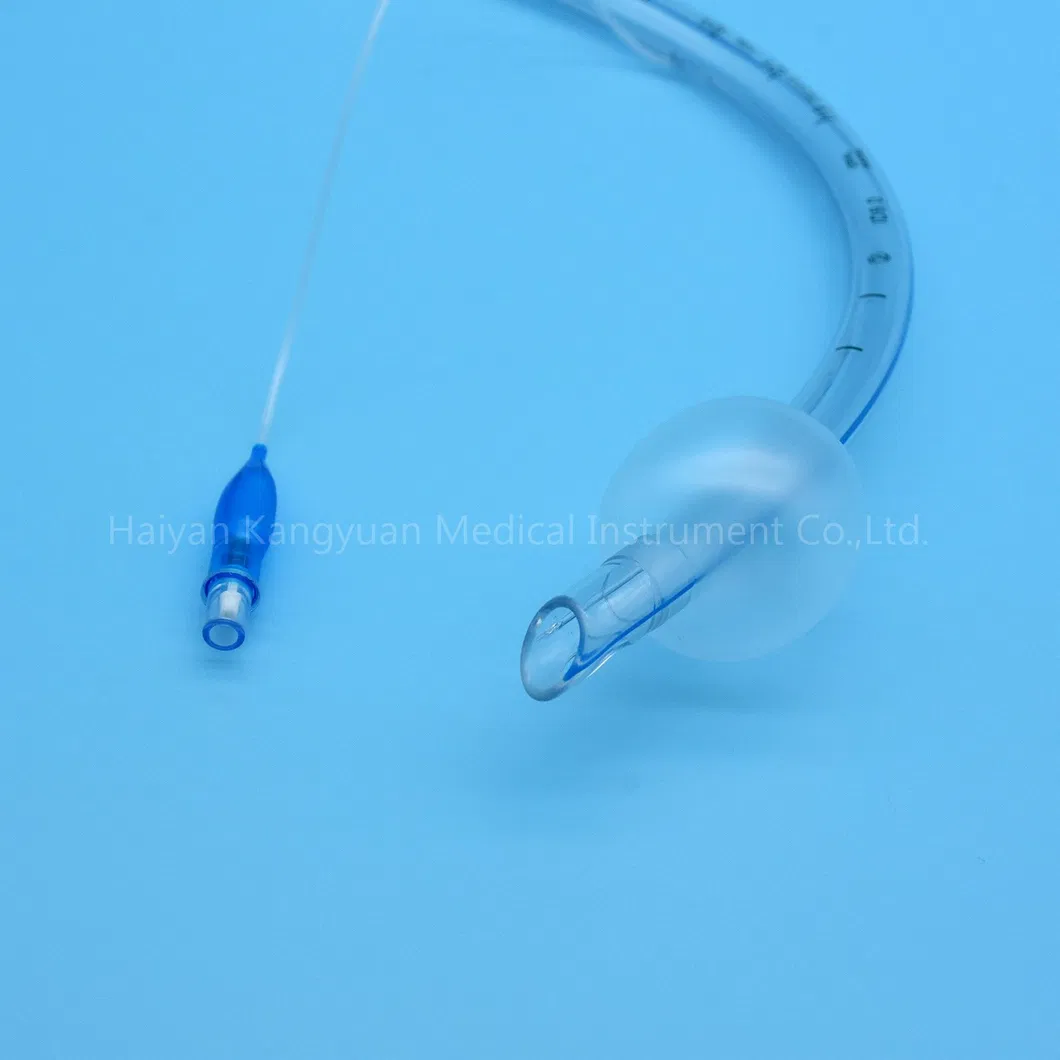
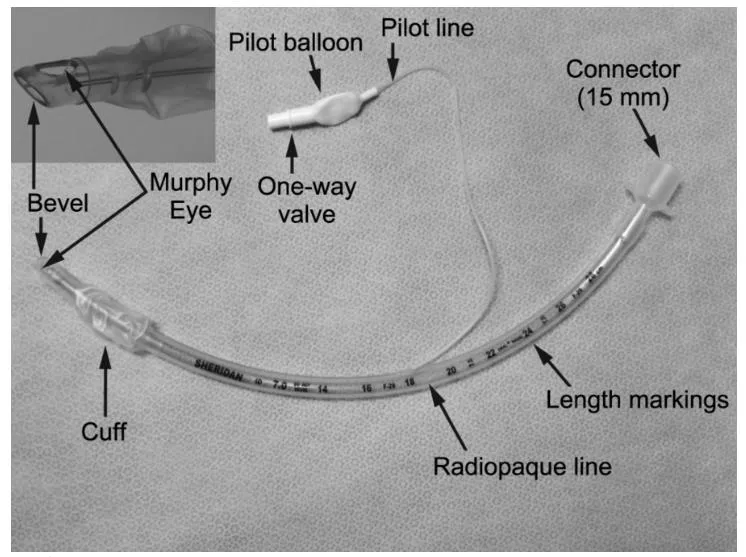
Enhanced Safety: The high volume low pressure cuff applies lower pressure against the tracheal wall, reducing the incidence of ischemia and necrosis.
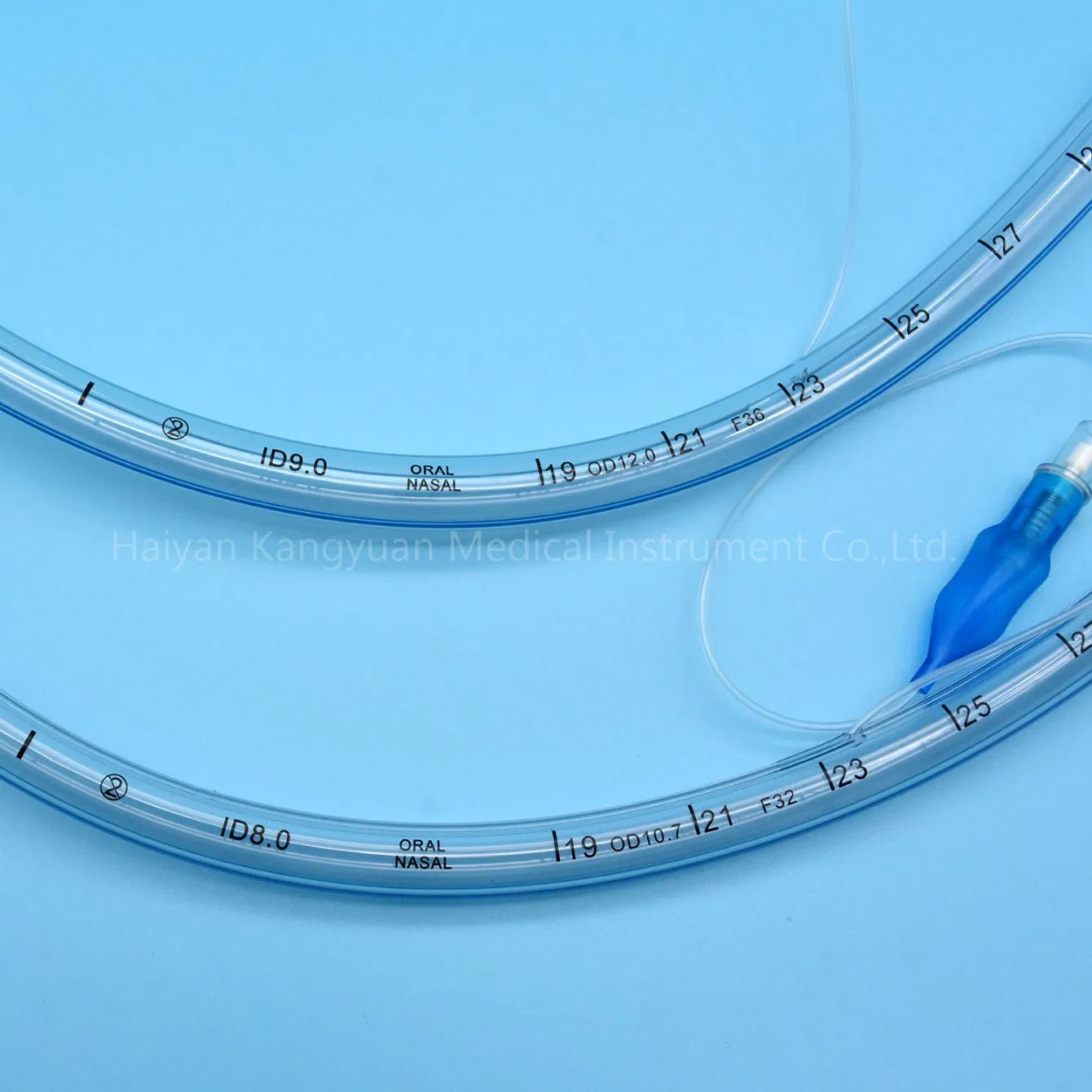
Clear Visualization: The radio-opaque line and pilot balloon provide critical feedback during and after intubation.
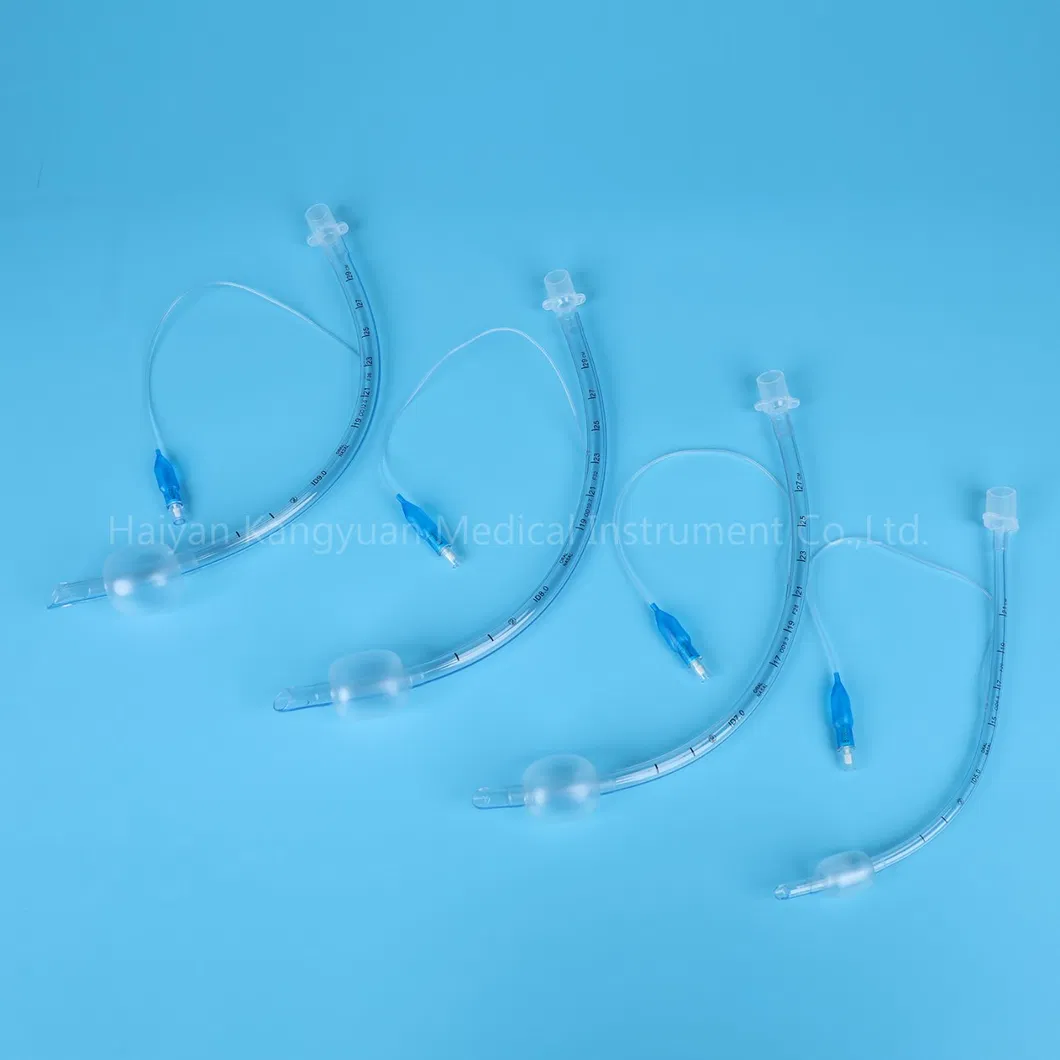
Versatile Use: Designed for both short and long-term intubations in hospital environments.
An endotracheal tube is a flexible tube placed through the mouth into the trachea (windpipe) to assist patient breathing. Connected to a ventilator, it delivers oxygen directly to the lungs. It remains the "gold standard" for securing and protecting the airway during surgery, trauma, or serious illness.


 Wigivida Medical
Wigivida Medical