




1 / 5










| Still deciding? Get samples of $ ! US$ 3.5/Piece Request Sample |
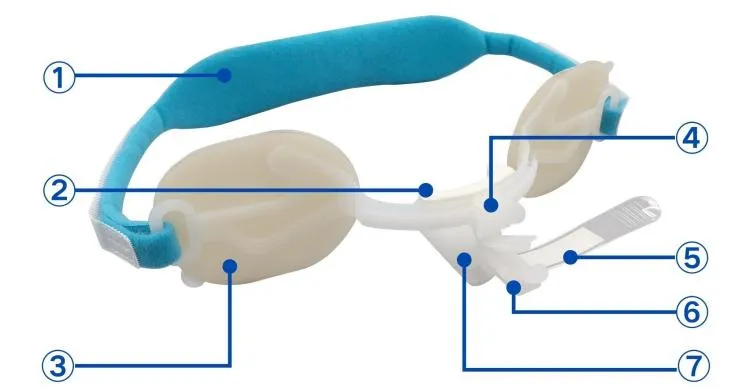
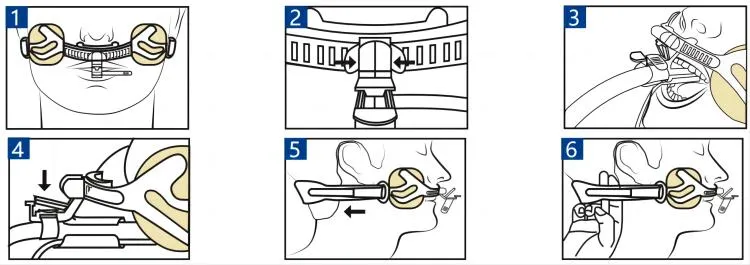
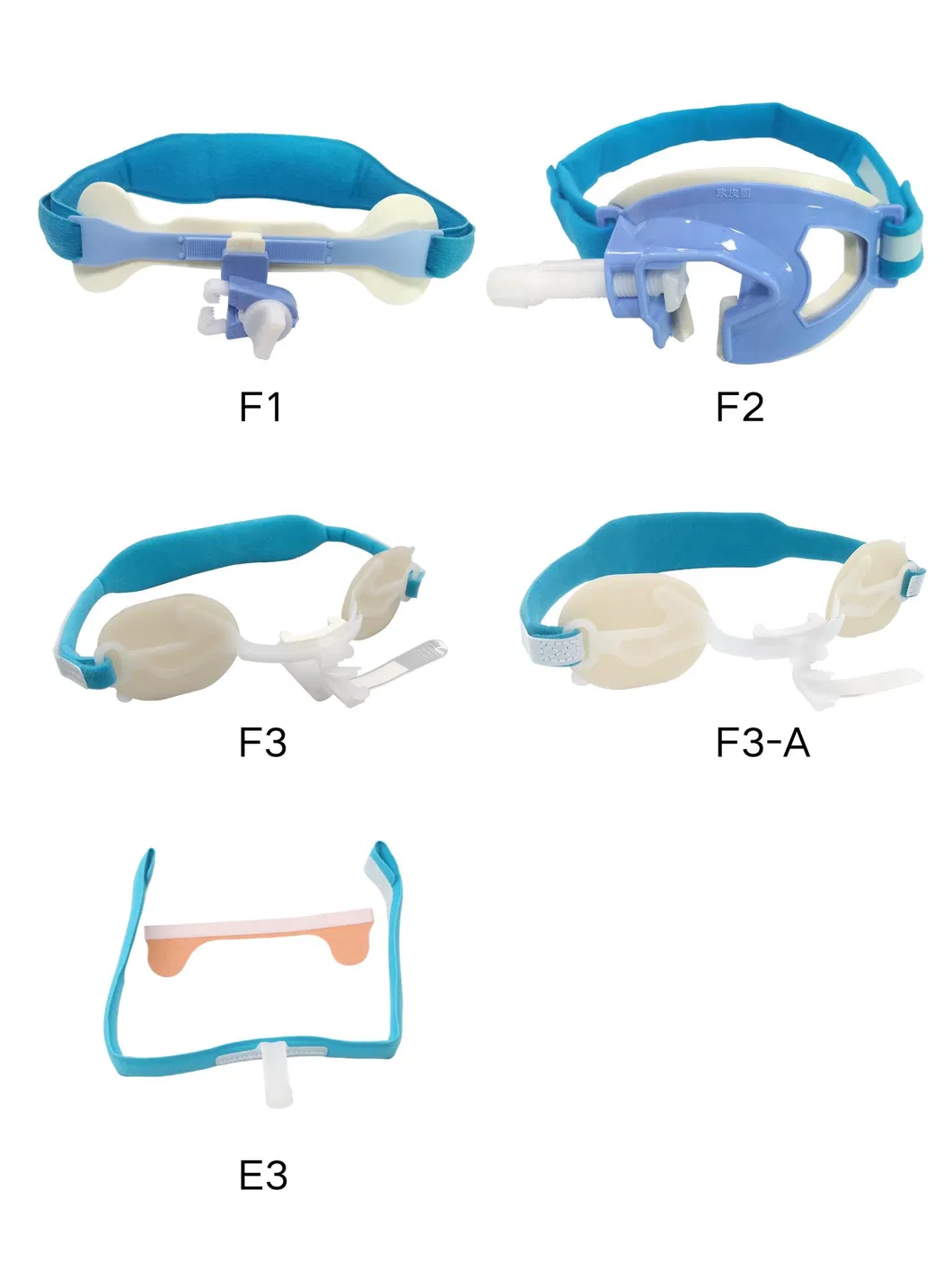
| Type | Code | Pcs/inner box | Pcs/master carton |
|---|---|---|---|
| F | F1 | 1 | 20 |
| F | F2 | 1 | 20 |
| F | F3 | 1 | 50 |
| F | F3-A | 1 | 50 |
| E | E3 | 1 | 100 |


The facility is a professional medical technology enterprise approved by governmental health authorities. Established with significant investment in foreign advanced production equipment, the company maintains a scientific management system. We specialize in the development, production, and sale of medical catheter fixation devices, transparent dressings, and fixation bands, holding certifications for Class I and Class II medical devices.


 Wigivida Medical
Wigivida Medical