














| Still deciding? Get samples of $ ! US$ 0.1/Piece Request Sample |
| PRODUCT SPECIFICATION | |
|---|---|
| Type | Hypodermic needle |
| Material | Stainless steel |
| Size | 15G, 16G, 18G, 20G, 21G, 22G, 23G, 25G, 26G, 27G, 29G, 30G |
| Use | Used with syringe or infusion set |
| Package | Blister packing |
| Application | Medical |
| Feature | Disposable |
| Certification | CE, ISO, GMP |
| Delivery time | 20 days |
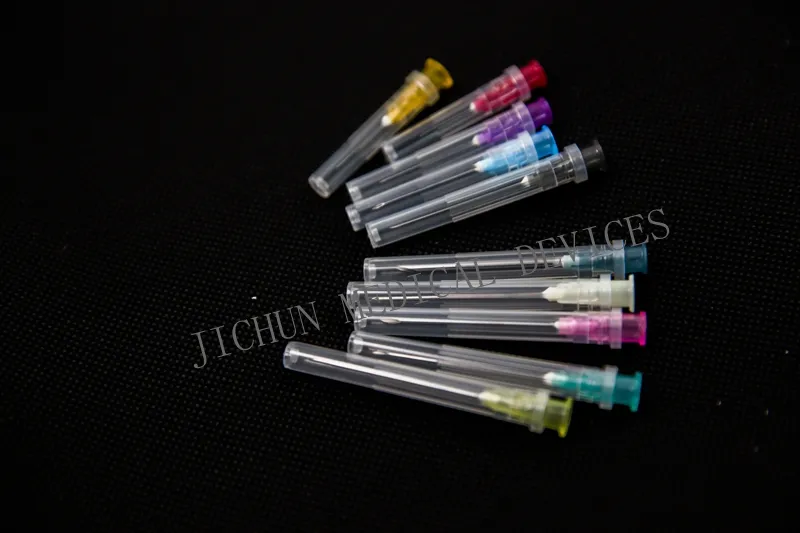
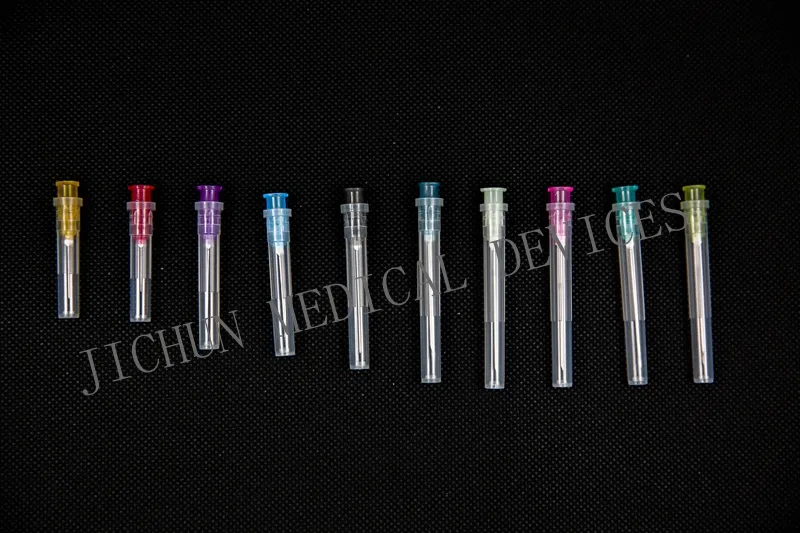
| Size | Color Code | Regular Needle Length | ||||||
|---|---|---|---|---|---|---|---|---|
| O.D | Gauge | 1/2" | 5/8" | 3/4" | 1" | 1 ¼" | 1 ½" | |
| 0.3 | 30G | Yellow | √ | |||||
| 0.33 | 29G | Red | √ | |||||
| 0.4 | 27G | Grey | √ | |||||
| 0.5 | 25G | Orange | √ | √ | √ | |||
| 0.6 | 23G | Blue | √ | √ | √ | |||
| 0.8 | 21G | Green | √ | √ | √ | |||
| 1.2 | 18G | Pink | √ | √ | ||||
| 1.8 | 15G | Dark blue | √ | |||||


Established in 1988, the manufacturer operates within a 40,000 square meter facility, including an 18,000 square meter purification plant. As a leading enterprise in the medical device industry, the company maintains strong technical expertise and advanced automated production equipment.
Currently, the annual production capacity reaches 2 billion disposable syringes, 60 million intravenous infusion needles, 1.8 billion sterile injection needles, and 100 million insulin pen needles. The company holds numerous patents, including 3 invention patents and 44 utility model patents, ensuring high standards in design and technical management.
The facility has achieved multiple international certifications including ISO9001, ISO13485, and CE0197. Recognized as a national high-tech enterprise, it continues to innovate through technological and systemic advancements to contribute to global human health. Products are distributed worldwide across Europe, America, Asia, Africa, and the Middle East.




 Wigivida Medical
Wigivida Medical