









| Still deciding? Get samples of $ ! US$ 0.01/Piece Request Sample |
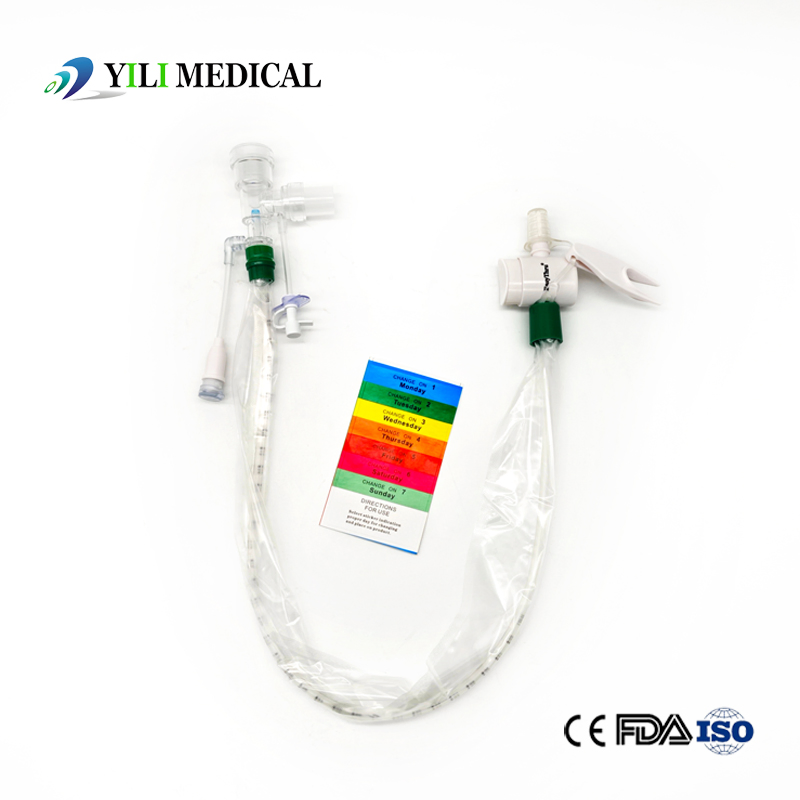
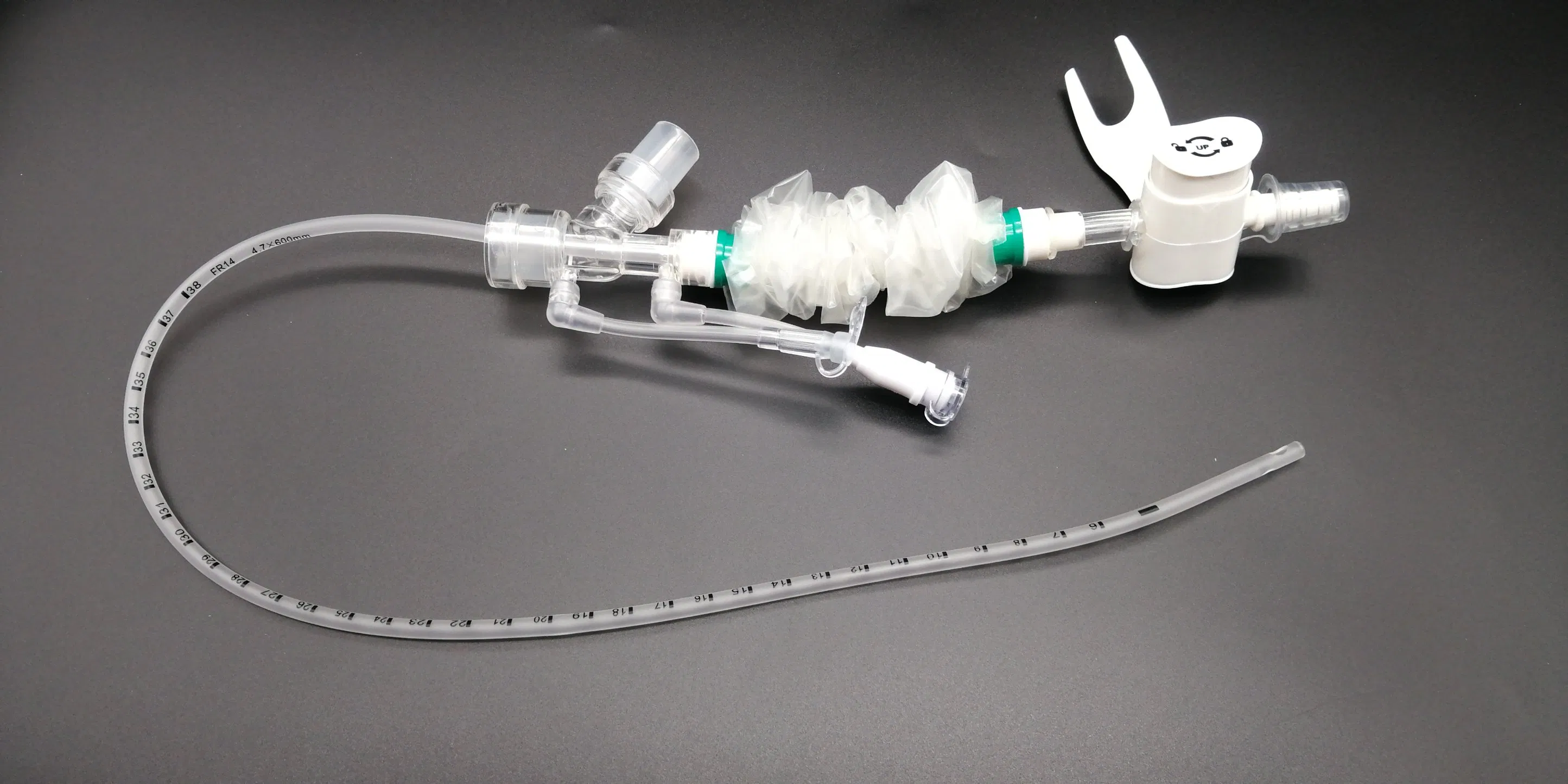
Surgical Disposable PVC Medical Suction Tube Catheter with 72 Hour Mark
What is Closed Suction System?
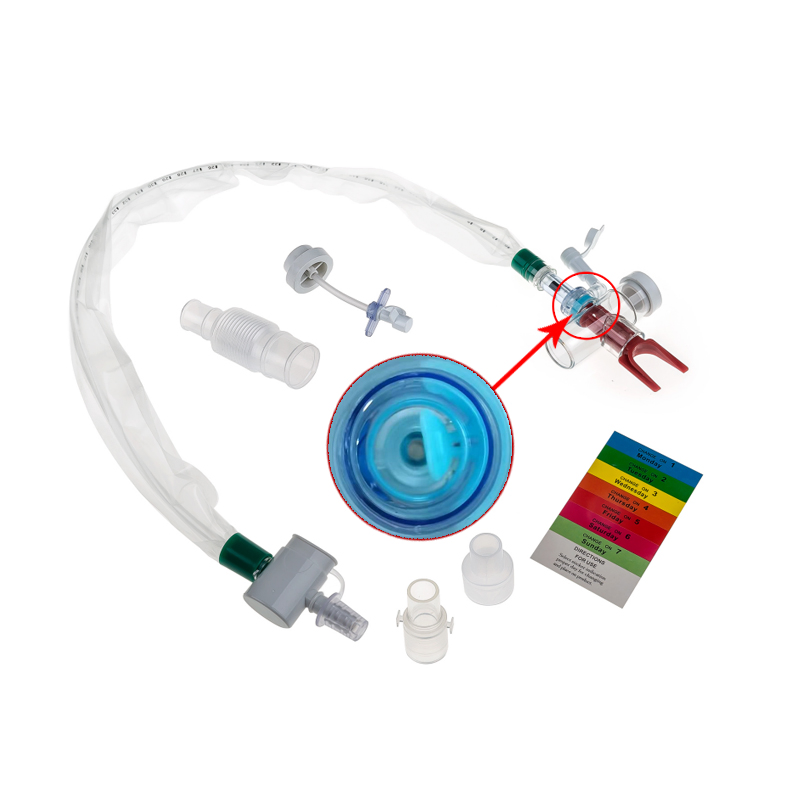
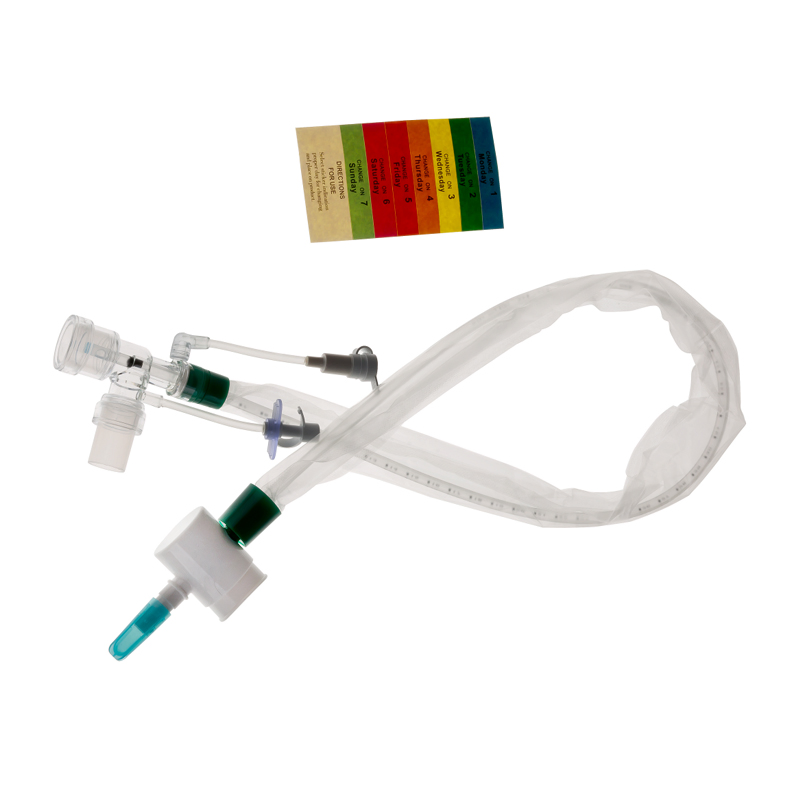
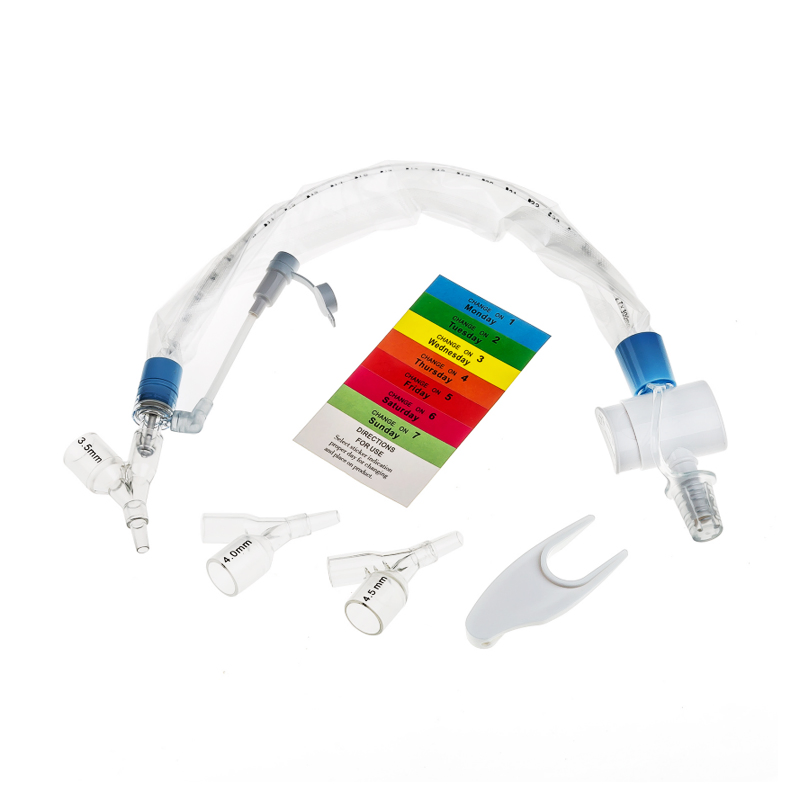
The Closed Suction System is intended for removal of secretion in the lower respiratory track in patients receiving prolonged mechanical ventilation. The filter protected system is connected to the artificial airway of the patient (Endotracheal or Tracheostomy tube) and to the ventilation circuit. It is used over a period of 24 hours or 72 hours depending on the patient requirement.
Product size range: Fr6, Fr8, Fr10, Fr12, Fr14, Fr16. Y Connector Type available for 6, 7, 8 French sizes.
| Application | Personal Care |
| Properties | Medical Materials & Accessories |
| After-sale Service | Online technical support |
Why use closed suction system?
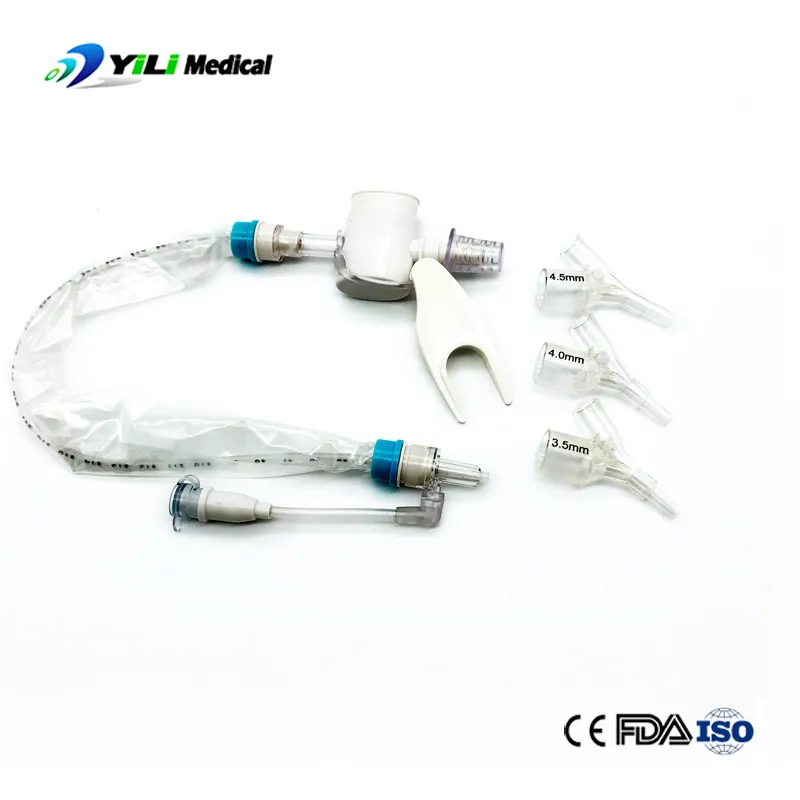
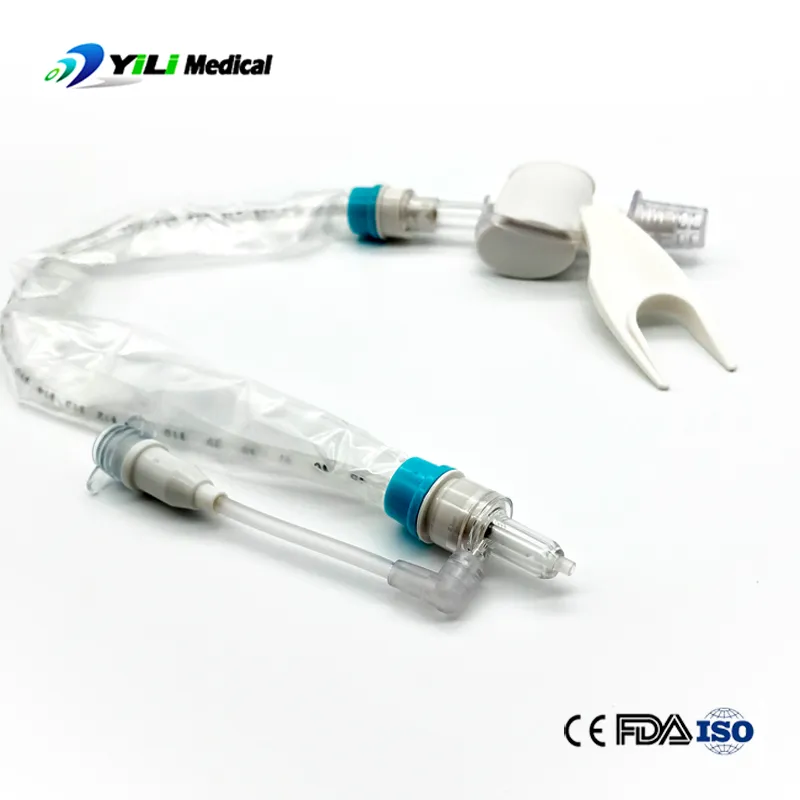
We are a professional manufacturer and supplier of medical supplies for over 9 years. Our main product lines include breathing anesthesia, urinary, and injection infusion products. Our facilities operate under strict quality control systems to ensure one-stop service and reliable product variety.


Product Qualification: Our factory runs under the ISO 13485 quality control system. Products are produced in Cleanrooms following strict SOPs to ensure every process is traceable. We provide finished goods testing reports before shipment.







 Wigivida Medical
Wigivida Medical