

1 / 5










| Still deciding? Get samples of $ ! US$ 0/Piece Request Sample |
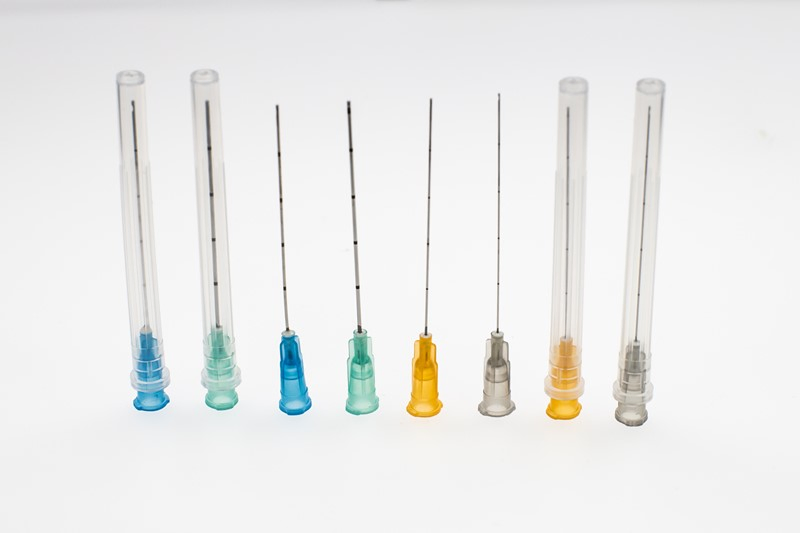
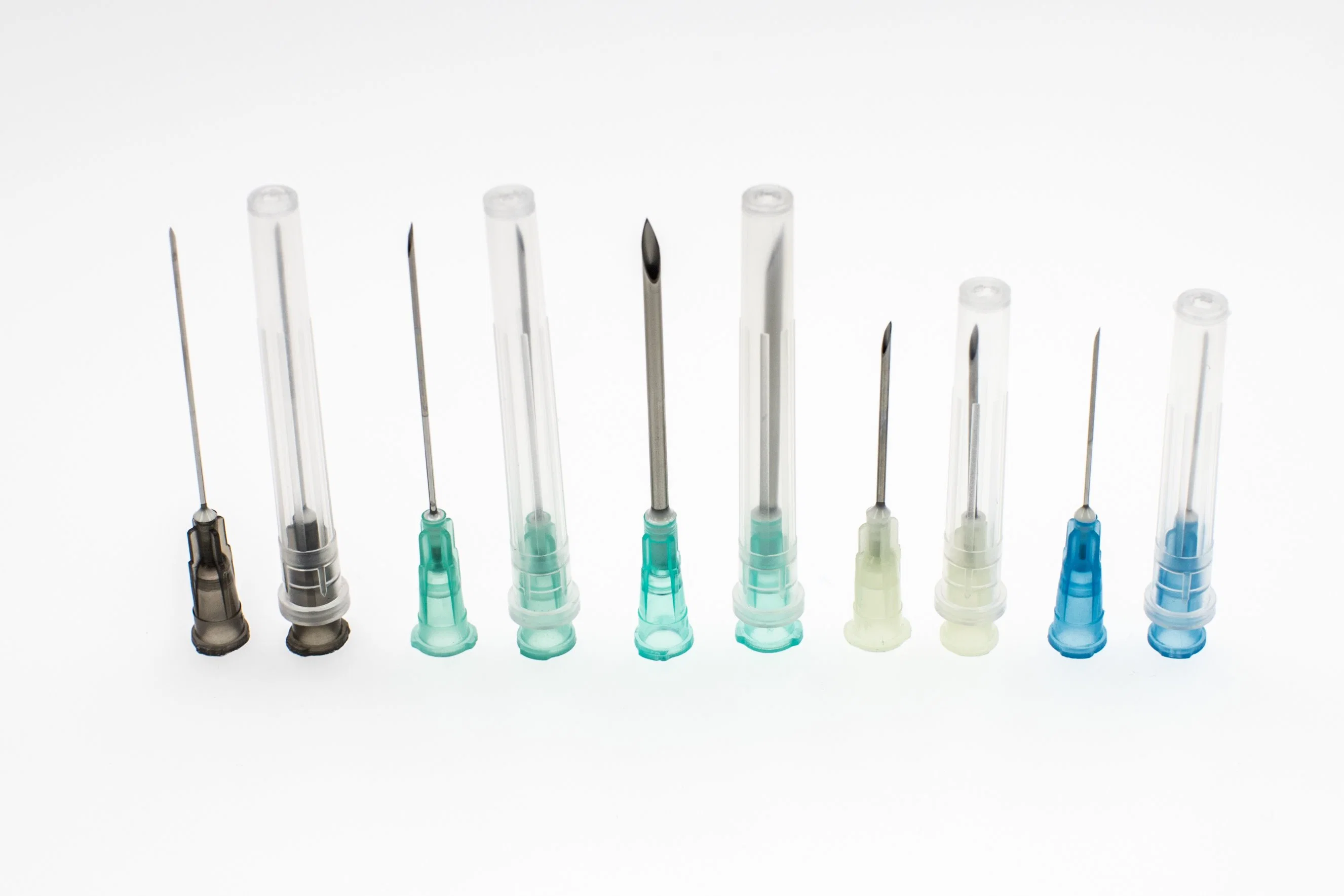
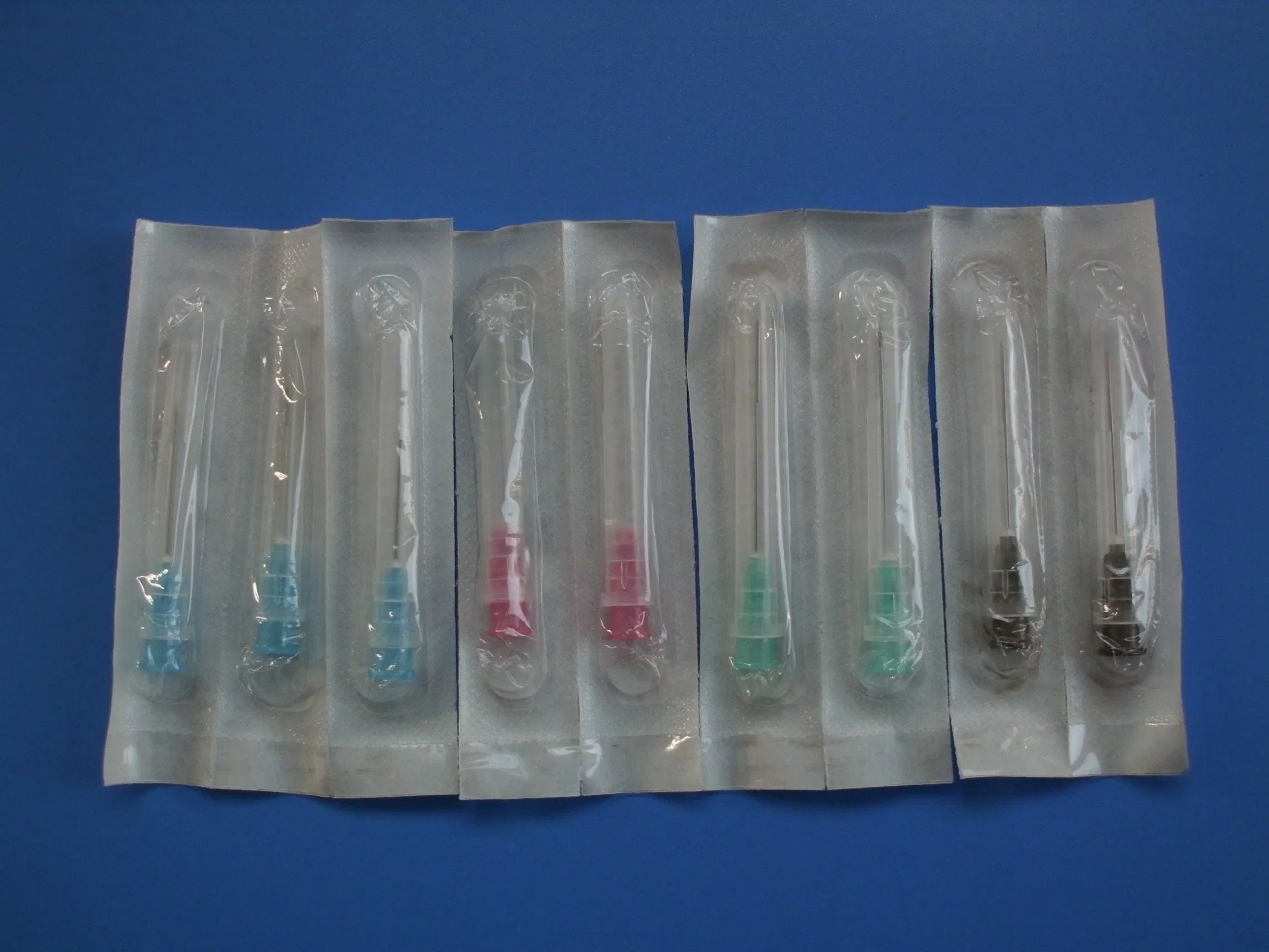
Disposable Sterile Hypodermic Medical Injection Syringe Needles
1. General Specifications:
2. Component Details:
| Component | Specifications & Materials |
|---|---|
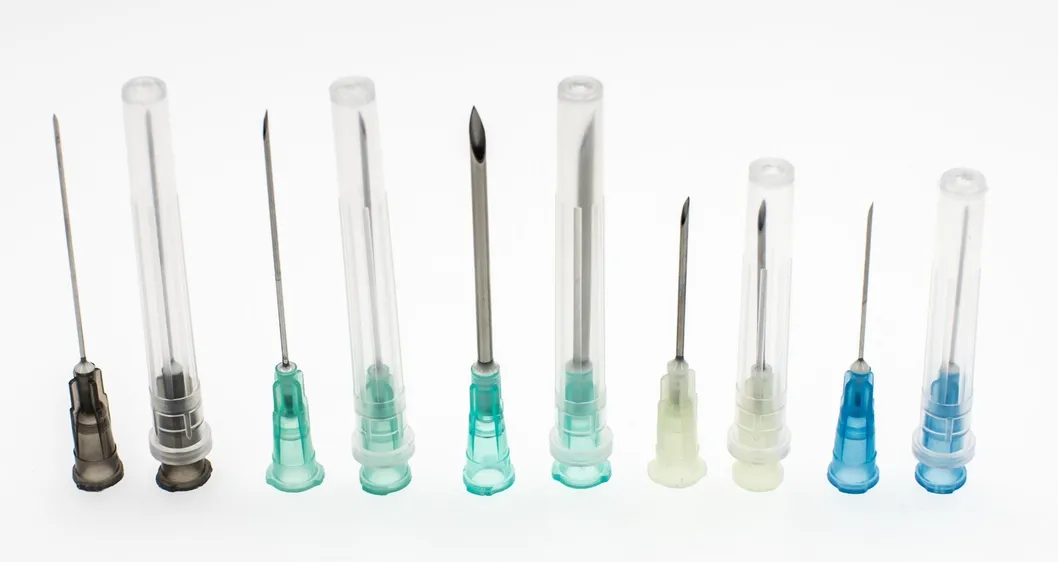
| Needle Cannula | Stainless steel AISI 304; Colored according to ISO 6009 |
| Needle Protector | Medical high transparent PP; Length matches needle size |
| Lubricant | Medical silicone (ISO 7864) |
| Sterilization | EO Gas; Non-toxic and non-pyrogenic |
| Applications | Human injection or Veterinary use |
| Certificates | CE, ISO13485 |


We are located in the economically developed Yangtze River Delta region and are committed to the research, development, and production of disposable sterile medical devices. Our core product range includes disposable syringes, infusion sets, vaginal dilators, urine bags, and tourniquets.
Our facilities have achieved EU SGS certification, ISO 13485, and ISO 9001 quality system certifications. All research, production, and sales processes are governed by a strict quality management assurance system, ensuring high standards through rigorous supervision and inspection.

 Wigivida Medical
Wigivida Medical