Engineered for precision and maximum protection, our top-tier safety needles meet the rigorous demands of Australian clinical environments.
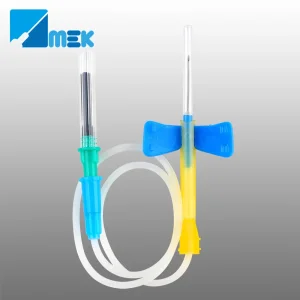
Australia's healthcare sector is globally recognized for its stringent safety standards and commitment to healthcare worker (HCW) protection. As a premier Safety Needles Factory and Supplier, we understand that the Australian market is driven by the Therapeutic Goods Administration (TGA) regulations and the AS/NZS 3816:2018 standards for management of clinical and related wastes.
In major hubs like Sydney, Melbourne, and Brisbane, public health networks such as NSW Health and Queensland Health have pioneered the mandatory adoption of Safety-Engineered Devices (SEDs). This shift is not merely a trend but a critical response to preventing Needle-Stick Injuries (NSIs), which historically posed significant risks of blood-borne pathogen transmission like Hepatitis B, C, and HIV.
Our role as a global supplier to the Australian market involves bridging the gap between high-volume manufacturing efficiency and the localized precision required by Australian pathology labs and oncology centers. Whether it's for Huber needles used in chemotherapy or butterfly needles for routine venipuncture, our products are designed to integrate seamlessly into the Australian clinical workflow.
Data reflects the growing demand for safety-engineered medical devices across Australian private and public healthcare sectors.
The Australian market is moving away from active safety (user-activated) to passive safety mechanisms where the needle is shielded automatically after use, minimizing human error.
With Australia's focus on sustainability, there is a rising demand for safety needles that use less plastic and are compatible with high-efficiency sharps containers.
Ultra-sharp, silicone-coated tips are now standard to ensure patient comfort, a key metric for private clinics in high-end Australian medical districts.
Smart packaging and QR-coded batch tracking are becoming essential for inventory management in large Australian hospital groups like Ramsay Health Care.

Our safety needles are engineered for diverse applications across the Australian continent:
Foshan Wigivida Medical Co., Ltd. is a leading China-based manufacturer of high-quality medical consumables and devices, specializing in respiratory products, medical tubes, urology products, hypodermic and surgical consumables. With years of dedicated experience in the healthcare industry, Wigivida has built a reputation for providing reliable, safe, and innovative solutions to hospitals, clinics, and medical distributors worldwide, including a strong presence in the Australian market.
The company offers a comprehensive product range, including oxygen masks, ventilator accessories, catheters, Foley catheters, hypodermic needles, syringes, surgical drapes, sterile gloves, sutures, cotton swabs, gauze rolls, IV sets, and disposable medical kits. Each product is manufactured with strict adherence to quality standards, ensuring biocompatibility, durability, and patient safety.

Wigivida Medical emphasizes innovation, research, and precision manufacturing, combining advanced production technology with a rigorous quality management system. The company also provides customized solutions to meet the specific needs of healthcare providers, supporting efficient hospital workflows and minimizing infection risks.
Guided by the principles of "quality first, service first, and customer satisfaction-driven," Foshan Wigivida Medical Co., Ltd. has earned an excellent reputation both domestically and internationally. With a professional R&D team, modern manufacturing facilities, and dedicated after-sales service, the company continues to advance the medical consumables industry, delivering safe, effective, and high-quality healthcare solutions that improve patient outcomes and support global healthcare innovation.
Our expertise as a global supplier is backed by rigorous testing and clinical validation.
Our design team includes clinical consultants who ensure that every safety mechanism—whether it's a sliding sleeve or a spring-retraction system—is intuitive for nurses under pressure.
We maintain ISO 13485 certification and our products carry CE and FDA 510(k) clearances, aligning with the high expectations of the Australian TGA for imported medical devices.
We understand the logistical challenges of the Australian market. Our streamlined export processes ensure consistent supply to distributors in New South Wales, Victoria, and beyond.
Explore our extensive range of safety-engineered devices available for Australian medical distribution.
As the demand for high-quality medical consumables grows in Australia, Foshan Wigivida Medical Co., Ltd. remains committed to being the preferred Safety Needles Factory and Supplier. Our focus on patient safety, healthcare worker protection, and TGA-aligned quality makes us the ideal partner for Australian medical distributors and healthcare networks. By choosing our safety-engineered devices, you are investing in a future where needle-stick injuries are a thing of the past, and clinical excellence is the standard.