 Medical Disposable Yankauer Handle Set with Suction Connecting Connection Cannula Catheter Tube
Medical Disposable Yankauer Handle Set with Suction Connecting Connection Cannula Catheter Tube
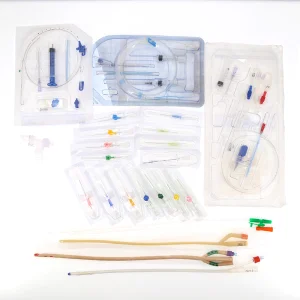 Disposable Medical PVC Latex Silicone Foley Closed Suction Central Venouse Hemodialysis Dialysis IV Cannula Urethral Catheter
Disposable Medical PVC Latex Silicone Foley Closed Suction Central Venouse Hemodialysis Dialysis IV Cannula Urethral Catheter
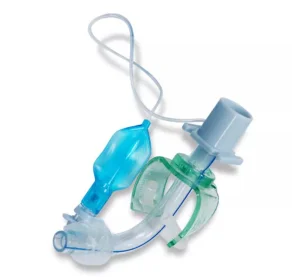 Factory Price Single-Use PVC Cuffed Uncuffed Tracheostomy Tube with Inner Cannula Suction Port Disposable Tracheostomy Tube
Factory Price Single-Use PVC Cuffed Uncuffed Tracheostomy Tube with Inner Cannula Suction Port Disposable Tracheostomy Tube
 Safety Multi-Sample Test Sampling Blood Collection Pen Type Blood Needle with FDA
Safety Multi-Sample Test Sampling Blood Collection Pen Type Blood Needle with FDA
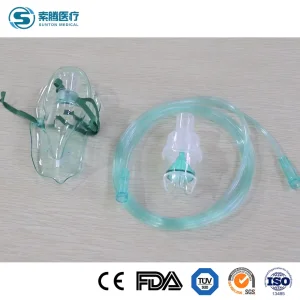 Portable Audlt/Pediatric/Infant Disposable Medical Oxygen Nasal Cannula Nebulizer Aerosal Face Facial Mask with Connecting Tubing with CE/ISO
Portable Audlt/Pediatric/Infant Disposable Medical Oxygen Nasal Cannula Nebulizer Aerosal Face Facial Mask with Connecting Tubing with CE/ISO
 Disposable Multi Sample Pen Type Flashback Safety Blood Collection Needle with Holder
Disposable Multi Sample Pen Type Flashback Safety Blood Collection Needle with Holder
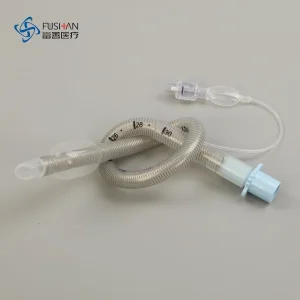 OEM ODM Silicone Medical Supply Disposable Endotracheal Tube Cuffed Uncuffed Anaesthesia PVC Tracheal Tracheostomy Cannula CE ISO Cfda Certified Ett 3.0-10.0mm
OEM ODM Silicone Medical Supply Disposable Endotracheal Tube Cuffed Uncuffed Anaesthesia PVC Tracheal Tracheostomy Cannula CE ISO Cfda Certified Ett 3.0-10.0mm
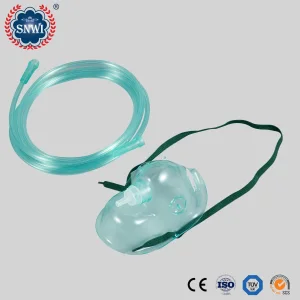 CE ISO Approved Hospital Disposable Sterile Medical PVC Oxygen Mask/Nebulizer Mask/Venturi Mask/Non Rebreathing Oxygen Mask with Reservoir Bag
CE ISO Approved Hospital Disposable Sterile Medical PVC Oxygen Mask/Nebulizer Mask/Venturi Mask/Non Rebreathing Oxygen Mask with Reservoir Bag
The global Pen Type IV Cannula market is undergoing a significant transformation, driven by a universal emphasis on patient comfort, clinician safety, and the optimization of infusion therapy. As an essential component in clinical settings, the IV cannula (Intravenous Cannula) is the lifeline for delivering fluids, medications, and blood products directly into the circulatory system. In today's industrialized medical sector, the "Pen Type" design remains one of the most widely utilized configurations due to its ergonomic grip, ease of insertion, and reliability in emergency and routine procedures alike.
Industrial data indicates a robust CAGR (Compound Annual Growth Rate) in the demand for peripheral intravenous catheters (PIVCs). This growth is attributed to the increasing prevalence of chronic diseases, rising surgical procedures worldwide, and a burgeoning aging population that requires frequent medical intervention. As a leading China Pen Type IV Cannula manufacturer, Foshan Wigivida Medical Co., Ltd. sits at the intersection of this global demand, providing high-precision instruments that meet stringent international regulatory standards such as CE, ISO 13485, and FDA guidelines.
The industry trend is shifting toward "Safety IV Cannulas" to prevent needle-stick injuries (NSIs). Our Pen Type models feature optimized needle bevels for atraumatic insertion and options for automatic retraction technology, significantly reducing the risk for healthcare workers.
Modern IV cannulas utilize Medical Grade FEP (Fluorinated Ethylene Propylene) or Polyurethane (PUR). These materials are kink-resistant and soften once inside the vein, minimizing the risk of phlebitis and increasing indwelling time.
Following international standards (ISO 10555-5), our manufacturing line ensures strict color-coding for gauge sizes—from 14G (Orange) for rapid transfusion to 26G (Violet) for neonates, ensuring error-free selection in high-pressure environments.
SEO Insight: When searching for a reliable IV cannula supplier, global procurement officers prioritize manufacturers who demonstrate E-E-A-T (Experience, Expertise, Authoritativeness, and Trustworthiness). At Wigivida, our transparency in material sourcing and our rigorous multi-stage QC process (Visual inspection, Leakage testing, and Sterility verification) establish us as a top-tier authority in the China medical export market.
China's integrated medical industrial clusters allow us to source high-grade stainless steel and polymers at scale, reducing lead times for massive global orders.
From custom packaging and branding to specific needle lengths or catheter materials, our R&D team provides end-to-to-end customization to suit local market requirements in Europe, Africa, or the Americas.
Our products undergo rigorous testing to ensure compliance with CE and ISO certifications, making them ready for immediate distribution in most international markets.
Automated production lines minimize human error while maintaining a cost structure that allows hospitals and distributors to optimize their procurement budgets without sacrificing safety.
The versatility of the Pen Type IV Cannula makes it indispensable across various medical settings:

Modern medical procurement is no longer just about the lowest price. B2B buyers from medical distribution hubs in Dubai, Germany, and the USA are looking for a "Total Quality Package." This includes sterilization certificates, biocompatibility reports, and stable lead times. The increasing shift toward centralized government tenders in many regions requires suppliers who can handle volume without dipping in quality. Wigivida Medical addresses these needs by maintaining a buffer stock and utilizing ERP-driven manufacturing to track every batch from raw material to the final sterile pouch.
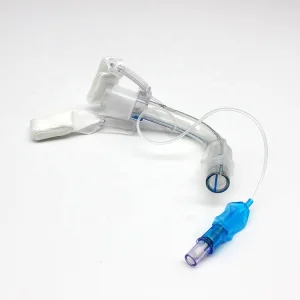 Disposable PVC Tracheostomy Tube Cuffed or Uncuffed Endotracheal Tracheostomy Tube with Inner Cannula
Disposable PVC Tracheostomy Tube Cuffed or Uncuffed Endotracheal Tracheostomy Tube with Inner Cannula
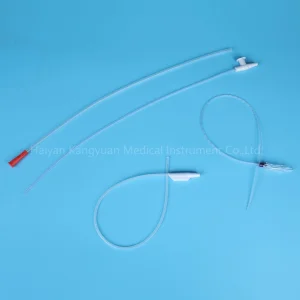 Suction System Catheter Aspiratory Tube Medical Device for Respiratory Treatment Oxygen PVC Factory China Wholesale Medical Tube Cannula
Suction System Catheter Aspiratory Tube Medical Device for Respiratory Treatment Oxygen PVC Factory China Wholesale Medical Tube Cannula
 Premium Medical Grade Pen-Style Safety IV Indwelling Needle with Automatic Retraction Technology & Sterile Single-Use Design
Premium Medical Grade Pen-Style Safety IV Indwelling Needle with Automatic Retraction Technology & Sterile Single-Use Design
 PVC PP Disposable Hypodermic Needle & Cannula Sterile Syringe Needle/Stainless Steel Hypodermic Needle for Medical Use
PVC PP Disposable Hypodermic Needle & Cannula Sterile Syringe Needle/Stainless Steel Hypodermic Needle for Medical Use
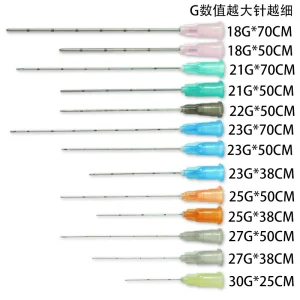 CE FDA Micro Cannula Blunt Tip with Hypodermic Needle for Filler Dermal Filler Cannulas Safety Blunt Tip Cannula
CE FDA Micro Cannula Blunt Tip with Hypodermic Needle for Filler Dermal Filler Cannulas Safety Blunt Tip Cannula
 21g Pen Type Safety Blood Collection Needle with CE ISO
21g Pen Type Safety Blood Collection Needle with CE ISO
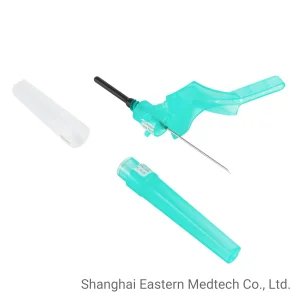 Vaccine Medical Products Hospital Instruments New Version Safety Blood Collection Needle Pen Type
Vaccine Medical Products Hospital Instruments New Version Safety Blood Collection Needle Pen Type
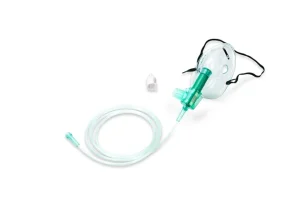 Medical Grade PVC Nasal Oxygen Cannula Oxygen Mask Nebulizer Mask with Tube
Medical Grade PVC Nasal Oxygen Cannula Oxygen Mask Nebulizer Mask with Tube
Foshan Wigivida Medical Co., Ltd. is a leading China-based manufacturer of high-quality medical consumables and devices, specializing in respiratory products, medical tubes, urology products, hypodermic and surgical consumables. With years of dedicated experience in the healthcare industry, Wigivida has built a reputation for providing reliable, safe, and innovative solutions to hospitals, clinics, and medical distributors worldwide.

The company offers a comprehensive product range, including oxygen masks, ventilator accessories, catheters, Foley catheters, hypodermic needles, syringes, surgical drapes, sterile gloves, sutures, cotton swabs, gauze rolls, IV sets, and disposable medical kits. Each product is manufactured with strict adherence to quality standards, ensuring biocompatibility, durability, and patient safety.
Wigivida Medical emphasizes innovation, research, and precision manufacturing, combining advanced production technology with a rigorous quality management system. The company also provides customized solutions to meet the specific needs of healthcare providers, supporting efficient hospital workflows and minimizing infection risks. Guided by the principles of "quality first, service first, and customer satisfaction-driven," Foshan Wigivida Medical Co., Ltd. has earned an excellent reputation both domestically and internationally. With a professional R&D team, modern manufacturing facilities, and dedicated after-sales service, the company continues to advance the medical consumables industry, delivering safe, effective, and high-quality healthcare solutions that improve patient outcomes and support global healthcare innovation.